 Grai Orthopedic Implants
Grai Orthopedic Implants
 Grai Orthopedic Implants
Grai Orthopedic Implants
When addressing the crucial topic of orthopedic surgery, understanding how to check if orthopedic implants are compatible with surgical instruments is essential. Compatibility directly affects the success of surgeries and patient outcomes. Surgeons rely on a seamless interaction between implants and instruments for optimal performance.
A series of steps must be taken to ensure that the instruments can effectively work with the implants. This process involves evaluating dimensions, materials, and design features. For instance, mismatched sizes can lead to complications during procedures. Inadequate checks can result in delays and increased risks in the operating room.
Developing a thorough checklist for compatibility is vital. This checklist should include defined protocols and expert evaluations. Orthopedic surgeons should collaborate with manufacturers to obtain accurate specifications. Confirming compatibility not only guarantees a smoother surgical experience but also fosters patient safety and trust in healthcare systems.
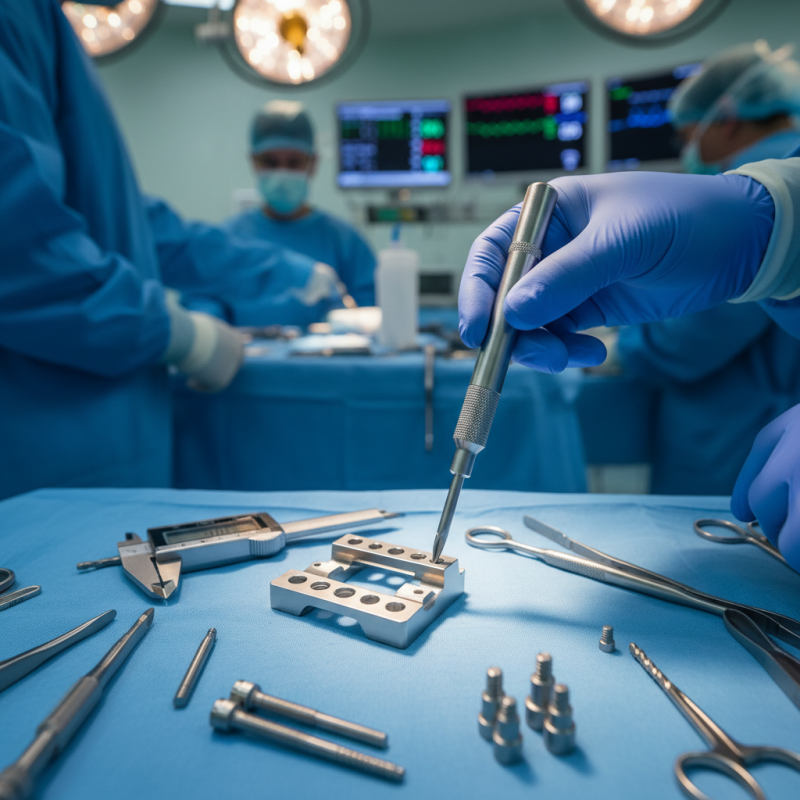
Orthopedic implants play a crucial role in modern medicine. They support healing and restore function in damaged bones and joints. These devices come in various shapes and materials. Each type is designed to fit specific anatomical needs. Understanding their functionality is essential for healthcare professionals. The right implant can significantly influence a patient's recovery and quality of life.
Evaluating compatibility with instruments is important in orthopedic surgery. Different instruments may not work well with certain implants. For example, a surgical tool may not align correctly with an implant's design. This misalignment can hinder the operation. Surgeons must assess both the implants and the instruments thoroughly. A lack of compatibility could lead to complications during surgery.
Choosing the correct implant requires careful consideration. Every patient's anatomy is unique. Therefore, a one-size-fits-all approach does not apply. Surgeons should reflect on their experiences and adapt their choices. Continual learning is vital in this evolving field. As new technologies emerge, so do new challenges. A flexible mindset will help healthcare providers navigate these complexities effectively.
Orthopedic implant compatibility testing involves various instruments that assess how well these devices interact with each other. Key tools include deformation gauges and axial load testing machines. According to a recent report by the Orthopedic Industry Association, about 30% of implant failures are linked to compatibility issues. Using precise instruments can help identify these potential failures early in the design process.
Deformation gauges measure changes in dimensions when implants are subjected to stress. This testing helps determine if the implant can endure the forces it will face in the body.
Axial load testing machines apply force along the length of the implant. They reveal how well the implant will perform under real-world conditions. These tests are vital. Yet, one must remember that results can vary due to material properties and manufacturing techniques.
Additionally, understanding the biomechanical environment is crucial. Each instrument has its limitations. For instance, some gauges may not measure tiny deformations accurately. This can lead to oversight. Hence, a combined approach using multiple testing instruments often yields more reliable data.
Implementing stringent compatibility testing could enhance patient outcomes and reduce revision surgeries. However, continuous innovation in testing methods is still needed to ensure the highest standards.
Checking the compatibility of orthopedic implants with instruments is vital for successful surgeries. Begin by evaluating the size and design of both the implant and the instruments used. Clinical data indicates that misalignment can lead to complications, affecting up to 10% of procedures (Orthopedic Clinical Studies Journal, 2022).
Next, assess the materials used in the implant and instruments. Compatibility in materials can influence the longevity and performance of the surgical outcome. Research shows that certain materials can interact negatively, increasing the risk of infection or failure. Recent studies reveal that around 15% of implant failures stem from poor material choices (International Orthopedic Research Journal, 2023). It is crucial for medical professionals to cross-reference material safety data sheets.
Lastly, conduct trials in a controlled environment, if possible. Observing the interaction of implants and instruments under simulated conditions can yield insights. However, not all scenarios can be replicated accurately in a lab. The real-world complexities can challenge predictions made during these tests. Continuous education and updated data are essential for improving compatibility assessments.
Evaluating the compatibility of orthopedic implants with surgical instruments poses several challenges. One major challenge is the variability in manufacturing standards. Different manufacturers may use distinct materials and design specifications. This leads to concerns about how well implants function with certain instruments during surgery. If the instruments do not match the dimensions or mechanics of the implant, complications may arise.
Another issue is the lack of comprehensive testing. Often, compatibility is assessed through limited clinical trials. This means that results may not accurately reflect the implants' performance in diverse surgical environments. Surgeons might face unexpected difficulties when using instruments that were not thoroughly evaluated for specific implants. It's crucial to address these gaps in testing, as they can impact patient safety and surgical outcomes.
Surgeons must also navigate the complexity of surgical procedures. When instruments are incompatible, procedures may take longer, leading to increased risk for patients. The communication among surgical teams about potential incompatibilities is vital. Regular discussions and updates on new technologies can foster better understanding and adaptability. This ongoing dialogue is essential to improve surgical efficiency and patient care.
In orthopedic surgery, compatibility testing is crucial for ensuring surgical success and patient safety. According to a report from the Journal of Orthopedic Research, nearly 30% of implant failures are attributed to incompatibility with surgical instruments. When implants and instruments are mismatched, the risk of complications increases significantly. Surgeons need to assess materials, dimensions, and mechanical properties carefully to avoid these issues.
Testing compatibility helps clinicians identify potential failures before surgery. A study by the American Academy of Orthopaedic Surgeons (AAOS) found that proper alignment and fit can reduce operational complications by over 25%. Surgeons should consider how different implants interact with specific instruments during procedures. A mismatch can lead to delays, increased blood loss, and longer recovery times.
Despite technological advancements, challenges remain. Some instruments are not universally compatible with all implant types. Reflections on past errors help improve current practices. Enhanced training for surgical teams in identifying compatibility issues is vital. Overall, understanding compatibility's role can elevate surgical outcomes and patient experiences.
: Compatibility ensures successful surgeries and reduces complications, which can affect up to 10% of procedures.
Size, design, and materials of both the implant and instruments must be assessed thoroughly.
Poor material choices can increase the risk of infection or implant failure, affecting performance.
Research shows that around 15% of implant failures stem from poor material choices.
Yes, variability in manufacturing standards and limited clinical testing can complicate evaluations.
Misalignment can lead to complications and longer surgical times, increasing risks for patients.
Regular communication and updates on new technologies can enhance understanding and adaptability.
No, real-world complexities often cannot be replicated accurately in lab conditions.
Limited testing may not reflect actual performance in diverse surgical environments.
Continuous education and updated data are essential for improving compatibility assessments.
In the realm of orthopedic surgery, ensuring the compatibility of implants with surgical instruments is crucial for successful patient outcomes. To understand "how to check if orthopedic implants are compatible with surgical instruments," it is essential to first comprehend the functionality of various orthopedic implants and identify the key instruments utilized during compatibility testing. A systematic approach involves a step-by-step assessment process to evaluate both implant and instrument interaction, while also addressing common challenges that may arise during compatibility evaluations.
Furthermore, the importance of compatibility testing cannot be overstated, as it plays a significant role in ensuring surgical success and patient safety. By thoroughly assessing the compatibility of orthopedic implants with the necessary surgical instruments, medical professionals can mitigate risks and improve overall surgical efficacy. This comprehensive understanding and methodical approach contribute significantly to the advancement of orthopedic procedures and patient care.
