 Grai Orthopedic Implants
Grai Orthopedic Implants
 Grai Orthopedic Implants
Grai Orthopedic Implants
 Hot Sale Intramedullary Nails System Medical Orthopedic Trauma Implant Surgery Pfna Proximal Femoral Intramedullary Interlocking Nail Pfna
Hot Sale Intramedullary Nails System Medical Orthopedic Trauma Implant Surgery Pfna Proximal Femoral Intramedullary Interlocking Nail Pfna
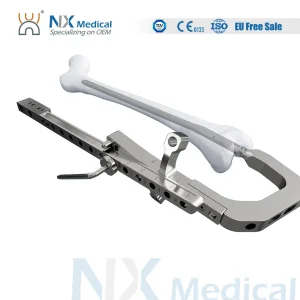 Nx Medical Retrograde Titanium Femoral Intramedullary Interlocking Nail System for Orthopedic Implants
Nx Medical Retrograde Titanium Femoral Intramedullary Interlocking Nail System for Orthopedic Implants
 Nx Medical Orthopedic Retrograde Femoral Intramedullary Interlocking Nail System for Trauma Implant
Nx Medical Orthopedic Retrograde Femoral Intramedullary Interlocking Nail System for Trauma Implant
 Manual Hip Joint Femoral Instrument Set Standard Stainless Instrument Total Knee Joint Replacement Instrument Set Hip and Knee
Manual Hip Joint Femoral Instrument Set Standard Stainless Instrument Total Knee Joint Replacement Instrument Set Hip and Knee
 Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
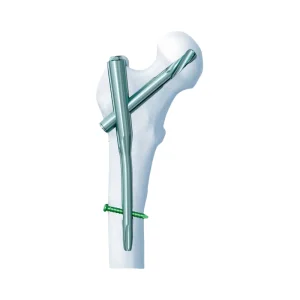 Gama Medical Surgical Bone System Pfn Titanium Orthopedic Trauma Surgery Femoral Intramedullary Implants Pfna Proximal Femur Interlocking Nail
Gama Medical Surgical Bone System Pfn Titanium Orthopedic Trauma Surgery Femoral Intramedullary Implants Pfna Proximal Femur Interlocking Nail
 Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement
Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement
 Factory Price Quality Assured Bone Surgery Orthopedic Surgical Instruments Pfna Femoral Interlocking Intramedullary Nails Instrument Sets
Factory Price Quality Assured Bone Surgery Orthopedic Surgical Instruments Pfna Femoral Interlocking Intramedullary Nails Instrument Sets
In the contemporary medical device industry, Femoral Interlocking Nails have become the "gold standard" for treating long bone diaphyseal fractures. As population demographics shift towards an aging society in developed nations and urbanization accelerates in emerging economies, the demand for high-performance intramedullary fixation has reached unprecedented heights. Globally, the orthopedic trauma market is witnessing a compound annual growth rate (CAGR) of over 6%, driven largely by high-velocity trauma cases such as road traffic accidents and geriatric fragility fractures.
Leading China Wholesale Femoral Interlocking Nails Manufacturers have bridged the gap between premium quality and cost-effectiveness. The industrial status today is defined by a shift from simple stainless steel implants to high-biocompatibility Titanium Alloy (Ti-6Al-4V ELI). These materials offer a superior strength-to-weight ratio and a modulus of elasticity closer to human bone, which significantly reduces the "stress shielding" effect. As a primary Femoral Interlocking Nail Factory, we are at the forefront of this evolution, ensuring that our products meet the rigorous standards of international healthcare systems.
Transitioning from Grade 5 Titanium to specialized surface treatments like anodic oxidation to enhance osteointegration and reduce infection risks.
New PFNA (Proximal Femoral Nail Antirotation) designs allow for smaller incisions, reduced blood loss, and faster post-operative recovery.
Integration with 3D surgical planning software, allowing surgeons to select the perfect nail length and diameter before the first cut.
Future trends point towards "Smart Implants" equipped with sensors to monitor bone healing and load distribution. Furthermore, the rise of 3D-printed instruments and customized nail systems for complex revision surgeries is reshaping the industry. As a leading manufacturer, we invest 15% of our annual revenue into R&D to ensure our Femoral Interlocking Nails remain at the cutting edge of biomechanical engineering.
When international hospitals and medical device distributors look for a Wholesale Femoral Interlocking Nail Manufacturer, their requirements go beyond just the physical product. Key procurement factors include:
Always verify the raw material source. Premium manufacturers use medical-grade titanium that meets ASTM F136 standards. Ask for the Material Traceability Identification report before placing large-scale wholesale orders.
As a specialized Orthopedic Trauma Implant Factory, we offer a unique blend of massive production capacity and boutique-level precision. Our manufacturing facility is optimized for the high-precision machining required for interlocking mechanisms.
Factory Stats & Quality Commitment:
Factory covers an area of 15,000 m², certified with ISO13485, and BSCI. Our products are certified with CE, MDR, and FDA. We employ over 500 professionals, including 8 senior engineers and 10 QC specialists.
We provide Minor Customization, full Raw Material Traceability Identification, and rigorous Semi-product and finished product inspection. Our advanced laboratory facilities conduct comprehensive fatigue and static testing to ensure every nail can withstand the physiological loads of the human body.
Over 300 different models available. 10 new products introduced every month by our experienced R&D team. Customizable designs and packaging.
Strict processes and raw material traceability. Advanced laboratory facilities for comprehensive testing to meet MDR standards.
Free replacement within one year. Comprehensive after-sales support with agency policy assistance for global partners.


Our Femoral Interlocking Nails are designed for diverse clinical scenarios across the globe:
| Clinical Scenario | Recommended Nail Type | Key Benefit |
|---|---|---|
| Proximal Femoral Fractures (Elderly) | PFNA (Antirotation) | Better stability in osteoporotic bone. |
| Mid-shaft Diaphyseal Fractures | Standard Universal Nail | High torsional and bending strength. |
| Distal Femoral Fractures | Retrograde Femoral Nail | Preserves proximal anatomy, easier access. |
| Polytrauma Cases | Titanium Interlocking System | Rapid fixation, MRI compatibility. |
Whether in a high-tech trauma center in Berlin or a busy municipal hospital in Sao Paulo, our nails provide the reliability surgeons need. The Interlocking Intramedullary Nail system ensures rotational stability, preventing leg shortening and malunion, which are critical for restoring the patient's mobility and quality of life.
 Orthopedic Intramedullary Nail System Interlocking Nails Implant Factory Price CE Approved
Orthopedic Intramedullary Nail System Interlocking Nails Implant Factory Price CE Approved
 Medical Titanium Bone Surgical System Pfna Orthopedic Femoral Interlocking Intramedullary Nail
Medical Titanium Bone Surgical System Pfna Orthopedic Femoral Interlocking Intramedullary Nail
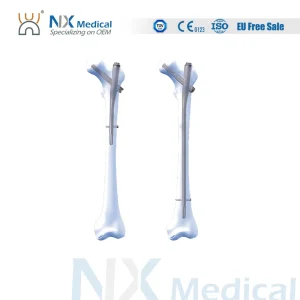 Nx Medical Orthopedic Implant Proximal Femoral Interlocking Intramedullary Nail
Nx Medical Orthopedic Implant Proximal Femoral Interlocking Intramedullary Nail
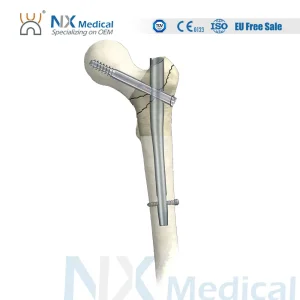 Nx Medical Orthopedic Trauma Titanium Intramedullary Interlocking Femoral Femur Pfna Bone Nail
Nx Medical Orthopedic Trauma Titanium Intramedullary Interlocking Femoral Femur Pfna Bone Nail
 Orthopedic Instruments Expert Tibial Interlocking Nails Instruments Set
Orthopedic Instruments Expert Tibial Interlocking Nails Instruments Set
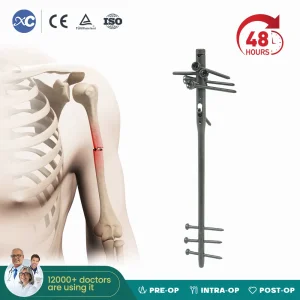 Xc Medico Orthopedic Implant Titanium Interlocking Humerus Nail Multi-Lock Humeral Intramedullary Nail
Xc Medico Orthopedic Implant Titanium Interlocking Humerus Nail Multi-Lock Humeral Intramedullary Nail
 Variable Angle Orthopedic LCP Distal Femoral Locking Plate
Variable Angle Orthopedic LCP Distal Femoral Locking Plate
 Orthopedic Fixation with Retrograde Intramedullary Femur Interlocking Intramedullary Nails 11*200
Orthopedic Fixation with Retrograde Intramedullary Femur Interlocking Intramedullary Nails 11*200