 Grai Orthopedic Implants
Grai Orthopedic Implants
 Grai Orthopedic Implants
Grai Orthopedic Implants

Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement

Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Total Knee Joint Replacement Instrument Set


Veterinary Hip Prosthesis Supplier Total Hip Joint Replacement Orthopaedic Surgery Implant and Tool

S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
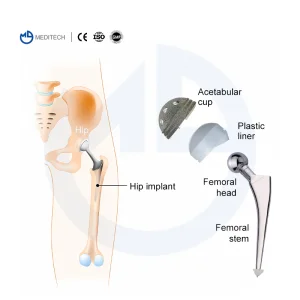
The global orthopedic market is witnessing a significant paradigm shift. As the average life expectancy increases worldwide, the demand for primary hip arthroplasty has surged over the past two decades. However, this has led to a subsequent rise in the need for Revision Hip Prosthesis. Revision surgery is a more complex procedure than primary replacement, requiring specialized implants that can account for bone loss, compromised tissue, and previous surgical trauma. As a leading OEM/ODM manufacturer, we understand that the revision market is no longer a niche segment but a vital pillar of the orthopedic industry.
Currently, the North American and European markets dominate the value share due to advanced healthcare infrastructure and high revision rates. Nevertheless, the Asia-Pacific region, led by China and India, is emerging as a massive growth engine. These markets are increasingly demanding high-quality, cost-effective revision solutions that can compete with premium Western brands while offering better accessibility.
Modern revision stems and acetabular cups are now utilizing additive manufacturing to create highly porous titanium surfaces. This mimics human trabecular bone, encouraging rapid osseointegration and providing superior stability in cases of severe bone loss.
The integration of AI and robotic navigation in revision surgery ensures precise implant placement. As a supplier, our instruments are designed to be compatible with latest digital planning softwares used by top-tier hospitals.
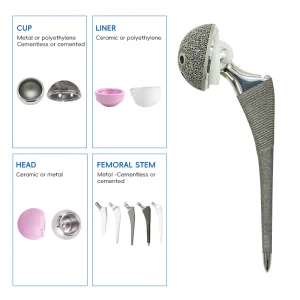
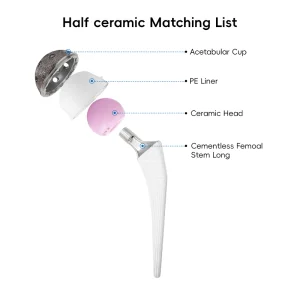
Modularity is key in revision hip systems (like our S-ROM compatible series). It allows surgeons to independently adjust offset, version, and length, providing a personalized fit for patients with complex anatomical challenges.
Beyond the hardware, the "biologics" trend is also influencing prosthesis design. Coatings such as Hydroxyapatite (HA) or antimicrobial surfaces are becoming standard requirements for global procurement. As an OEM partner, we stay at the forefront of material science, ensuring our titanium alloys and UHMWPE inserts meet the highest fatigue-testing protocols.
Procurement teams from international hospital groups, government health ministries, and private orthopedic distributors have distinct requirements. While quality is non-negotiable, the supply chain resilience and cost-to-value ratio have become critical factors. Our role as a China-based factory allows us to offer economies of scale without compromising on the stringent regulatory requirements like MDR or FDA clearance.
Localization Scenarios: In the Middle East and Southeast Asia, there is a high demand for "small-size" prosthesis suited for local anatomical dimensions. Conversely, in the Western market, "high-offset" and "long-stem" revision components are frequently requested. Our ODM services allow for these localized adjustments, providing customized surgical sets and implants that fit specific regional demographics.
With 8 senior engineers and a specialized R&D team, we produce over 300 different models. We introduce 10+ new products monthly to stay ahead of surgical trends.
Located in the heart of China's medical manufacturing hub, we optimize the production of Titanium and UHMWPE components, offering competitive pricing for global tenders.
Our facility is ISO13485 and BSCI certified. Products carry CE, MDR, and FDA marks, ensuring smooth entry into any international market.
Our Manufacturing Strength: Factory covers an area of 15,000 m², certified with ISO13485 and BSCI. We employ 500 professionals, including 8 senior engineers and 10 QC specialists. We offer minor customization, raw material traceability, and rigorous semi-finished and finished product inspections.
Innovative Solutions: Over 300 different models available. 10 new products introduced every month by our experienced R&D team. Customizable designs and packaging to meet your specific needs.
Customer Satisfaction: Free replacement within one year. After-sales support with agency policy assistance.


Revision hip replacement involves the removal of the old prosthesis and the insertion of a new one. This process is often necessitated by aseptic loosening, infection, periprosthetic fractures, or mechanical failure. Our Revision Hip Prosthesis systems are engineered to solve these challenges through:
As an industry-leading supplier, we don't just provide parts; we provide confidence. Our products, from the Corail-style femoral stems to the S-ROM titanium alloy joints, are tested under simulated physiological conditions to ensure long-term durability. Whether you are a veterinary hospital looking for small-dog hip replacements or a major medical center requiring human-grade implants, our OEM/ODM capabilities are designed to scale with your ambition.

Small Dog Joint Replacement Prosthesis 1#-5# a Type Total Hip Surgical Instrument


Primary Total Hip System Hip Joint Replacement Surgery Femoral Head

China Factory Orthopedic Implants Prosthetic Knee/Knee Joint Prosthesis/Artificial Knee Replacement



Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China

Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement