 Grai Orthopedic Implants
Grai Orthopedic Implants
 Grai Orthopedic Implants
Grai Orthopedic Implants
 Medical OEM&ODM Titanium Cannulated Headless Compression Screw Hollow Orthopedic Implant
Medical OEM&ODM Titanium Cannulated Headless Compression Screw Hollow Orthopedic Implant
 Cannulated Headless Compression Herbert Bone Screw Set Titanium Locking Orthopedic
Cannulated Headless Compression Herbert Bone Screw Set Titanium Locking Orthopedic
 Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants
Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants
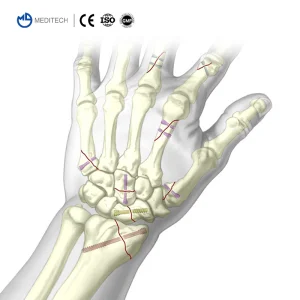 Titanium Medical Bone Screws Herbert Cannulated Compression Screw Price
Titanium Medical Bone Screws Herbert Cannulated Compression Screw Price
 Carbon Steel 4.8/8.8/10.9 Fastener Flower Countersunk Head Self-Tapping Screws
Carbon Steel 4.8/8.8/10.9 Fastener Flower Countersunk Head Self-Tapping Screws
 Orthopedic Cannulated Screw Half Threaded Locking Screw Titanium 7.3mm
Orthopedic Cannulated Screw Half Threaded Locking Screw Titanium 7.3mm
 SS304 Carbon Steel Knurled Head Thumb Screw
SS304 Carbon Steel Knurled Head Thumb Screw
 Customizable Steel Steel Black Finish Knurled Thumb Screw with Washer
Customizable Steel Steel Black Finish Knurled Thumb Screw with Washer
The global market for Cannulated Compression Screws is witnessing unprecedented growth, driven by a rising aging population and a surge in sports-related orthopedic injuries. As essential components in trauma surgery, these screws offer a minimally invasive approach to bone fixation, particularly in small bone fractures, femoral neck fractures, and arthrodesis.
Currently, the medical device industry is transitioning from traditional stainless steel to Grade 5 Titanium Alloys (Ti6Al4V ELI). This shift is prompted by the need for better biocompatibility, lower MRI interference, and superior fatigue resistance. As a leading exporter, we recognize that the North American and European markets maintain the highest demand for "headless" designs, which allow for intra-articular insertion without damaging surrounding soft tissue.
Our factory covers 15,000 m² and is fully certified with ISO13485 and BSCI. All orthopedic products carry CE, MDR, and FDA certifications, ensuring entry into the most regulated global markets.
With 500 employees, including 8 senior engineers, we introduce 10 new products monthly. Our R&D team focuses on the latest "Conical Headless" designs for maximum compression efficiency.
We provide full customization for medical brands, from minor design tweaks to complete private label packaging. Raw material traceability is guaranteed for every batch produced.
Sqm Factory Area
Available Models
Professional Staff
QC Inspection
The industry is moving beyond simple mechanical fixation. Today’s Cannulated Compression Screws are engineered with variable thread pitches to provide uniform compression across the fracture site. This "Herbert Screw" principle has been refined through advanced CNC machining, allowing for thinner walls and larger cannulation diameters to accommodate K-wires with higher precision.
For global procurement officers, the focus has shifted from "lowest price" to "total value." This includes raw material traceability, surface treatment quality (such as Type II anodization for titanium), and lead time reliability. Our factory leverages an integrated supply chain that ensures we can scale production rapidly while maintaining the strict tolerances required for medical-grade fasteners.
Foot & Ankle Surgery: Used extensively for Jones fractures and hallux valgus corrections.
Hand & Wrist Trauma: The gold standard for scaphoid non-union and small fragment fixation.
Pediatric Orthopedics: Ideal for epiphyseal injuries where minimal disruption is required.


Our strict quality control processes include semi-product and finished product inspections using advanced laboratory facilities. We offer a free replacement policy within one year for any quality concerns, backed by our robust after-sales support and agency policy assistance.
 Carbon Steel/4.8/8.8/10.9 M2.5 Galvanized Double Rod Fastener Knurled Bamboo Screw
Carbon Steel/4.8/8.8/10.9 M2.5 Galvanized Double Rod Fastener Knurled Bamboo Screw
 Orthopedic Implant Cannulated Headless Compression Screw - II Metallic Bone Screws
Orthopedic Implant Cannulated Headless Compression Screw - II Metallic Bone Screws
 Orthopedic Implant Cannulated Screw for CE Medical Grade Fixation
Orthopedic Implant Cannulated Screw for CE Medical Grade Fixation
 Orthopaedic Implant Cannulated Compression Screw 6.5 Cannulated Screw
Orthopaedic Implant Cannulated Compression Screw 6.5 Cannulated Screw
 Nx Medical Orthopedic Trauma Bone Implant Cannulated Conical Headless Compression Screw - II
Nx Medical Orthopedic Trauma Bone Implant Cannulated Conical Headless Compression Screw - II
 Nx Medical Orthopedic Cannulated Conical Headless Compression Screw - II for Bone Fixation
Nx Medical Orthopedic Cannulated Conical Headless Compression Screw - II for Bone Fixation
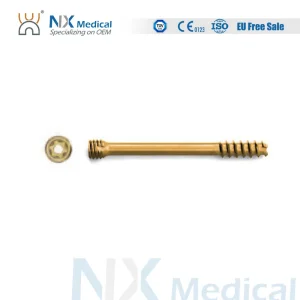 Nx Medical Orthopedic Implant Cannulated Headless Compression Screw Bone Fixation
Nx Medical Orthopedic Implant Cannulated Headless Compression Screw Bone Fixation
 Nx Medical Orthopedic Implant Cannulated Conical Headless Compression Screw - II Metallic
Nx Medical Orthopedic Implant Cannulated Conical Headless Compression Screw - II Metallic
When searching for a Top 10 Cannulated Compression Screws Factory, importers must look beyond the assembly line. A truly world-class manufacturer provides a comprehensive ecosystem of services. In the medical field, this means ensuring that every screw—whether it's a 2.5mm headless screw for wrist surgery or a 7.3mm cannulated screw for hip fractures—undergoes rigorous ultrasonic cleaning and is packaged in ISO Class 7 cleanrooms.
Our commitment to Innovative Solutions is evidenced by our massive catalog of over 300 different models. We understand that orthopedic trauma doesn't wait, which is why we maintain a high stock level of standard sizes while offering rapid prototyping for specialized OEM requests. By combining the cost-efficiency of Chinese manufacturing with international quality standards (MDR & FDA), we provide our global partners with a competitive edge that is hard to match.
As the industry evolves towards robotic-assisted surgery and smart implants, our engineering department is already exploring integrated sensor technologies and enhanced osteoconductive coatings. Partnering with us means securing your supply chain with the technology of tomorrow, available today.