 Grai Orthopedic Implants
Grai Orthopedic Implants
 Grai Orthopedic Implants
Grai Orthopedic Implants
 Medical Cement Spine Cannulated Pedicle Screw
Medical Cement Spine Cannulated Pedicle Screw
 Nx Medical Orthopedic Implant Mis Cannulated Bone Cement Spinal Pedicle Screw
Nx Medical Orthopedic Implant Mis Cannulated Bone Cement Spinal Pedicle Screw
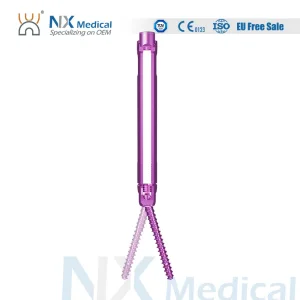 Nx Medical Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw 5.5mm System
Nx Medical Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw 5.5mm System
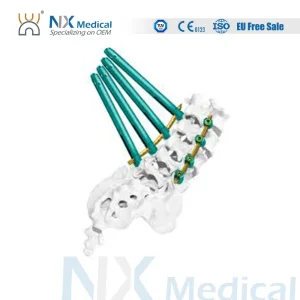 Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw 5.5mm System
Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw 5.5mm System
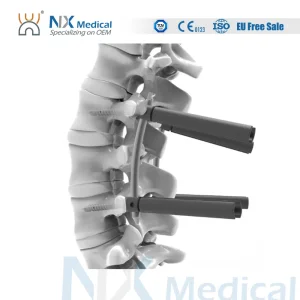 Nx Medical Orthopedic Implant Spine Cannulated Bone Cement Pedicle Screw Spinal Fixation
Nx Medical Orthopedic Implant Spine Cannulated Bone Cement Pedicle Screw Spinal Fixation
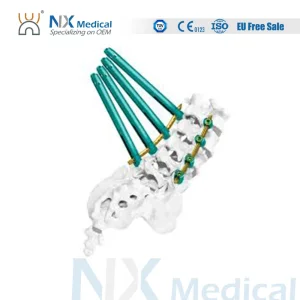 Nx Medical High-Quality Sf-IV Mis Cannulated Bone Cement Spinal Pedicle Screw 5.5mm
Nx Medical High-Quality Sf-IV Mis Cannulated Bone Cement Spinal Pedicle Screw 5.5mm
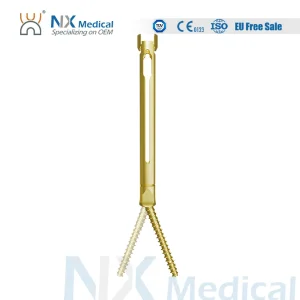 Nx Medical Titanium Cannulated Bone Cement Pedicle Screw 5.5mm Orthopedic Internal Fixation
Nx Medical Titanium Cannulated Bone Cement Pedicle Screw 5.5mm Orthopedic Internal Fixation
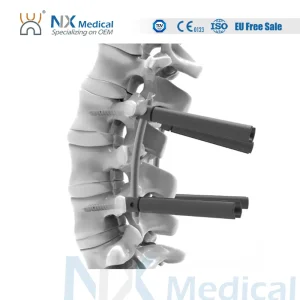 Nx Medical Titanium Cannulated Bone Cement 5.5mm System Pedicle Screw Bone Fixation
Nx Medical Titanium Cannulated Bone Cement 5.5mm System Pedicle Screw Bone Fixation
The global orthopedic market has witnessed a paradigm shift toward augmented fixation technologies. "Cement Spacers" and "Cannulated Bone Cement Screws" have become the gold standard for treating patients with poor bone quality, particularly in geriatric populations suffering from osteoporosis. As a leading exporter, we recognize that the integration of polymethylmethacrylate (PMMA) bone cement with titanium spinal systems is no longer a niche application but a global necessity.
Current industrial data suggests that the demand for spinal implants that support cement injection is growing at a CAGR of 7.5%. Hospitals and surgical centers in Europe, North America, and the Asia-Pacific region are increasingly prioritizing implants that offer immediate stability and reduced risk of screw loosening.

The shift from standard Grade 5 Titanium to bioactive coatings and advanced surface treatments that enhance osseointegration when combined with bone cement.
Modern bone cement spacers and cannulated screws are now designed to be compatible with robot-guided placement for sub-millimeter accuracy.
A move toward individual, sterile-packed implants (Ready-to-Use) to reduce hospital contamination risks and improve inventory management.
B2B Sourcing Requirements: Global distributors today require more than just a product; they need a partner capable of providing raw material traceability and comprehensive regulatory documentation (CE, FDA, MDR). Our cannulated bone cement systems meet these rigorous standards, ensuring smooth customs clearance and clinical acceptance in over 50 countries.
Local Application Scenarios:
✓ MDR & FDA Certification Compliance
✓ OEM/ODM Minor Customization
✓ 10 New Models Launched Monthly
✓ Direct-from-Factory Pricing

As a leading Chinese factory in the orthopedic sector, we leverage a robust supply chain and cutting-edge R&D to provide high-performance cement spacers and pedicle screws at competitive costs. Our factory covers an area of 15,000 m² and is certified with ISO13485 and BSCI.
With 500 employees, including 8 senior engineers and a dedicated 10-person QC team, we ensure that every cannulated screw undergoes strict quality control processes. From raw material traceability to finished product inspection in our advanced laboratory facilities, we guarantee reliability in every implant.
Innovative R&D: Our team introduces 10 new products every month, keeping our partners ahead of market trends. We offer customizable designs and packaging to meet specific localized needs across different healthcare systems.
Factory covers an area of 15,000 m², Factory certified with ISO13485, and BSCI, Products certified with CE, MDR, and FDA, 500 employees, including 8 senior engineers, and 10 QC, Minor Customization, Raw material traceability identification, Semi product and finished product inspection.
Strict quality control processes and raw material traceability. Advanced laboratory facilities for comprehensive testing.
Innovative Solutions: Over 300 different models available. 10 new products introduced every month by our experienced R&D team. Customizable designs and packaging to meet your specific needs.
Customer Satisfaction: Free replacement within one year. After-sales support with agency policy assistance.
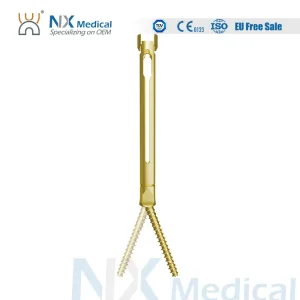 Nx Medical Titanium Cannulated Bone Cement Pedicle Screw Spine Internal Fixation
Nx Medical Titanium Cannulated Bone Cement Pedicle Screw Spine Internal Fixation
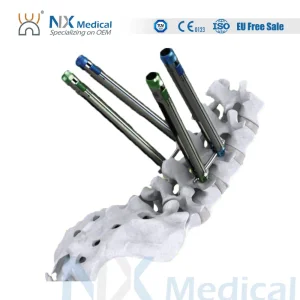 Nx Medical Orthopedic Implant Cannulated Bone Cement Pedicle Screw Spine Internal Fixation
Nx Medical Orthopedic Implant Cannulated Bone Cement Pedicle Screw Spine Internal Fixation
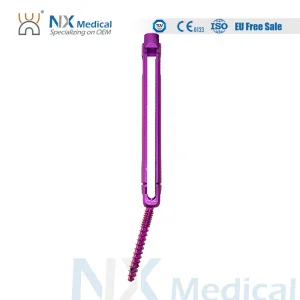 Nx Medical Titanium Cannulated Bone Cement Pedicle Screw Bone Internal OEM ODM
Nx Medical Titanium Cannulated Bone Cement Pedicle Screw Bone Internal OEM ODM
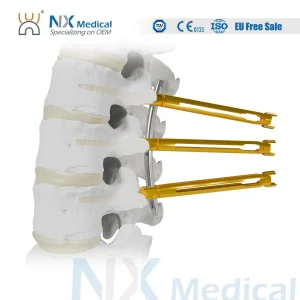 Nx Medical Spine Cannulated Bone Cement Pedicle Screw 5.5mm System Orthopedic Implant
Nx Medical Spine Cannulated Bone Cement Pedicle Screw 5.5mm System Orthopedic Implant
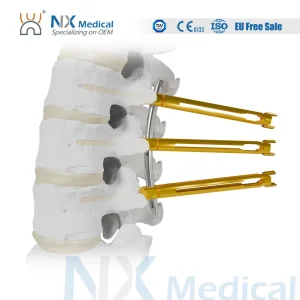 Nx Medical Orthopedic Sf-IV Mis Cannulated Bone Cement Spinal Pedicle Screw 5.5mm
Nx Medical Orthopedic Sf-IV Mis Cannulated Bone Cement Spinal Pedicle Screw 5.5mm
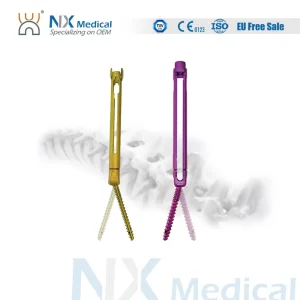 Nx Medical Titanium Cannulated Bone Cement Spinal Pedicle Screw Minimium Invasive
Nx Medical Titanium Cannulated Bone Cement Spinal Pedicle Screw Minimium Invasive
 Nx Medical OEM Support Titanium Cannulated Bone Cement Spinal Pedicle Screw
Nx Medical OEM Support Titanium Cannulated Bone Cement Spinal Pedicle Screw
 Nx Medical Titanium Cannulated Bone Cement Spinal Pedicle Screw Spine Internal Fixation
Nx Medical Titanium Cannulated Bone Cement Spinal Pedicle Screw Spine Internal Fixation