 Grai Orthopedic Implants
Grai Orthopedic Implants
 Grai Orthopedic Implants
Grai Orthopedic Implants
Jakarta, the heart of Indonesia's economic engine, has rapidly emerged as a sophisticated hub for Revision Hip/Knee Systems manufacturing. As the largest economy in Southeast Asia, Indonesia’s capital serves as a nexus for medical technology integration, combining localized industrial expertise with global orthopedic standards. The industrial landscape in Jakarta and its surrounding areas—including the strategic industrial corridors of Cikarang and Karawang—has shifted from basic medical supplies to high-precision implant engineering.
The demand for revision arthroplasty in the region is driven by a maturing patient population and an increase in primary joint replacement surgeries performed over the last decade. Our factory in Jakarta addresses the critical need for high-quality, biocompatible revision systems that can handle complex cases involving bone loss, infection, and mechanical failure. By leveraging Jakarta's proximity to world-class logistics and a highly skilled workforce, we ensure that specialized surgical tools and prostheses are available to healthcare providers across ASEAN.
Our Jakarta-based R&D teams are integrating AI-driven design protocols to create implants that better match the specific anatomical profiles of Indonesian and Asian patients, which often differ from Western averages.
Utilizing Medical-Grade Titanium and UHMWPE (Ultra-High Molecular Weight Polyethylene), we manufacture components that offer superior wear resistance and osseointegration, vital for revision success.
Factory certified with ISO13485 and BSCI. Our products meet CE, MDR, and FDA certifications, ensuring that Jakarta-made implants are globally competitive.
Being situated in Jakarta allows us to bypass global shipping bottlenecks, providing "Just-in-Time" delivery to local hospitals like RSCM and Siloam, reducing surgery wait times.
Our manufacturing philosophy in Jakarta centers on "Precision for Life." With a factory covering 15,000 m², we house state-of-the-art CNC machining centers, cleanroom packaging facilities, and an advanced laboratory for mechanical testing. We believe that a revision surgery is the patient's "second chance," and the tools used must be flawless.
R&D and Customization: We introduce over 10 new products every month. Our 8 senior engineers collaborate with top orthopedic surgeons in Jakarta to refine instrument sets, ensuring they are ergonomic and intuitive. Minor customizations for specific hospital protocols are standard practice for our team.
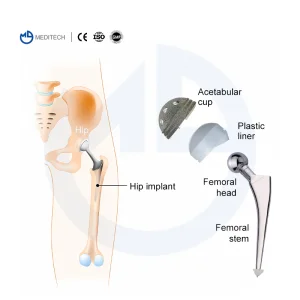
Employee Expertise: Our workforce of 500 includes specialized technicians and 10 dedicated Quality Control (QC) officers who manage raw material traceability from ingot to final sterilized package. This ensures that every component—be it a femoral stem or an acetabular cup—is documented and verified.


In the bustling medical ecosystem of Jakarta, Revision Arthroplasty is not just a procedure; it is a necessity for restoring mobility. Our revision systems are designed for several key localized application scenarios:
 Prosthesis Replacement Class III Knee Joint - Jakarta Edition
Prosthesis Replacement Class III Knee Joint - Jakarta Edition
 Jakarta Cemented Stem Hip Replacement & Revision Implant
Jakarta Cemented Stem Hip Replacement & Revision Implant
 Titanium Total Hip and Knee Joint Revision Systems
Titanium Total Hip and Knee Joint Revision Systems
 Corail Femoral Stem Titanium Prosthesis - Jakarta Specialized
Corail Femoral Stem Titanium Prosthesis - Jakarta Specialized
 S-ROM Titanium Alloy Cementless Hip for Revision Surgery
S-ROM Titanium Alloy Cementless Hip for Revision Surgery
 Jakarta Surgical Disposable Pulsed Lavage System
Jakarta Surgical Disposable Pulsed Lavage System
 Surgical DTN Retrograde Tibia Nail Revision Instrument
Surgical DTN Retrograde Tibia Nail Revision Instrument
 FNS Femoral Neck System Instrument Set - Jakarta Standard
FNS Femoral Neck System Instrument Set - Jakarta Standard
Our commitment in Jakarta extends beyond manufacturing. We offer a Free replacement policy within one year and comprehensive after-sales support including agency policy assistance. We recognize the importance of training and medical education in the successful deployment of Revision Hip/Knee Systems. As such, we partner with Jakarta’s medical associations to provide workshops and technical demonstrations for our advanced instrument sets.
Choosing a Jakarta-based manufacturer means choosing a partner who understands the local regulatory environment, the specific clinical challenges of Southeast Asian orthopedic surgery, and the need for rapid response times. Our 15,000m² facility is ready to serve global markets while maintaining the local agility that modern hospitals demand.