 Grai Orthopedic Implants
Grai Orthopedic Implants
 Grai Orthopedic Implants
Grai Orthopedic Implants
Sweden has long been recognized as a global leader in orthopedic innovation, largely due to the Swedish Hip Arthroplasty Register (SHAR), the world's first national quality registry for joint replacements. As a prominent Revision Hip/Knee Systems Supplier & Exporter, we align our product development with the rigorous clinical data and high-performance standards demanded by the Swedish medical community.
In Sweden, the demand for revision surgery—the procedure where a previous implant is replaced due to wear, infection, or instability—is evolving. With a population that remains highly active well into their senior years, Swedish orthopedic surgeons at institutions like Sahlgrenska University Hospital and the Karolinska Institute require revision systems that offer exceptional longevity and biological integration. The current trend in Sweden emphasizes bone conservation and modular design, allowing for personalized surgical approaches that adapt to the unique bone loss patterns of each patient.
Utilizing Grade 5 Titanium and advanced porous coatings to promote rapid osseointegration, essential for Swedish patients needing stable long-term fixation in revision scenarios.
Our R&D team utilizes computational modeling to simulate mechanical stress on Revision Hip stems, ensuring our exports to Sweden meet the highest durability benchmarks.
From Vitamin E infused UHMWPE liners to modular femoral stems, our systems are designed to minimize wear debris and prevent osteolysis, a key focus in Swedish clinical research.
The Swedish medical technology (MedTech) industry is currently undergoing a digital transformation. Robotic-assisted revision surgery is becoming more prevalent in major Swedish cities like Gothenburg and Uppsala. Our revision systems are designed with these navigational technologies in mind, featuring markers and geometries that are compatible with the latest surgical robots used in Nordic operating rooms.
Furthermore, sustainability is a core value in Sweden's industrial sector. As exporters, we provide eco-friendly sterilized packaging and follow BSCI-certified ethical manufacturing processes. This commitment to sustainability makes our Revision Hip/Knee systems highly attractive to Swedish public procurement agencies and private healthcare providers who prioritize ESG (Environmental, Social, and Governance) criteria.
Our factory covers an area of 15,000 m² and is fully certified with ISO13485 and BSCI. We take pride in our workforce of 500 employees, including 8 senior engineers and 10 QC specialists who ensure every product meets global medical standards.
With 10 new products introduced every month by our R&D team, we stay ahead of the curve in orthopedic innovation. We offer minor customization to suit specific anatomical needs often found in the Nordic population, ensuring that our Swedish partners receive the most effective tools for their patients.


 Prosthesis Replacement Sterilized Package Class III Knee Joint
Prosthesis Replacement Sterilized Package Class III Knee Joint
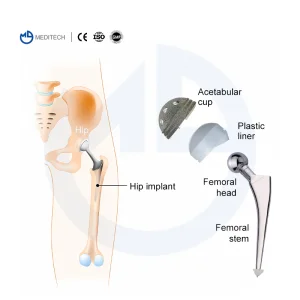 Cemented Stem Hip Replacement Sweden Application
Cemented Stem Hip Replacement Sweden Application
 Total Hip and Knee Joint Replacement Titanium Implant
Total Hip and Knee Joint Replacement Titanium Implant
 Total Hip Replacement Prosthesis Corail Femoral Stem
Total Hip Replacement Prosthesis Corail Femoral Stem
 S-ROM Titanium Alloy Cementless Hip Joints for Revision
S-ROM Titanium Alloy Cementless Hip Joints for Revision
 Disposable Pulsed Lavage System for Surgical Cleaning
Disposable Pulsed Lavage System for Surgical Cleaning
 Surgical Dtn Retrograde Tibia Nail Instrument
Surgical Dtn Retrograde Tibia Nail Instrument
 Fns Femoral Neck System Instrument Set
Fns Femoral Neck System Instrument Set
Revision hip and knee arthroplasty are significantly more complex than primary procedures. In the context of the Swedish healthcare system, where patient safety is paramount, selecting a supplier that understands the mechanical and biological nuances is crucial. Revision often involves bone loss, which requires specialized augmentations, such as wedges, blocks, and cones. Our systems provide a comprehensive modular platform that addresses these challenges effectively.
The S-ROM Titanium Alloy system, for instance, is highly favored for revision surgery due to its ability to provide proximal-distal mismatch solutions. This is particularly important for patients with varied femoral geometries. As exporters to Sweden, we ensure that our titanium implants undergo rigorous surface treatment to enhance "bone-on-growth," a critical factor for the long-term success of the implant in the absence of robust native bone.
Moreover, the rise of outpatient joint replacement in Nordic countries necessitates instruments and implants that allow for minimally invasive techniques even during revision. Our instrument sets, including the Fns Femoral Neck System and Retrograde Tibia Nail instruments, are optimized for surgical efficiency, reducing operating room time and accelerating patient recovery—a core goal of modern Swedish orthopedic protocols.
By partnering with us, Swedish medical distributors gain access to a supply chain that combines high-volume manufacturing efficiency with the precision of artisanal engineering. We are committed to supporting the Swedish orthopedic community in its mission to provide world-class care through reliable, innovative, and ethically manufactured revision systems.