



1 / 5
 Grai Orthopedic Implants
Grai Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 2.5/Piece Request Sample |
| Model NO. | WZ-TB-203 |
| Sterilization | Ethylene Oxide Sterilization |
| Quality Period | Two Years |
| Group | Adult |
| Usage | Single-Use |
| Application | Orthopedic Surgery |
| Origin | China |
| HS Code | 8108903290 |
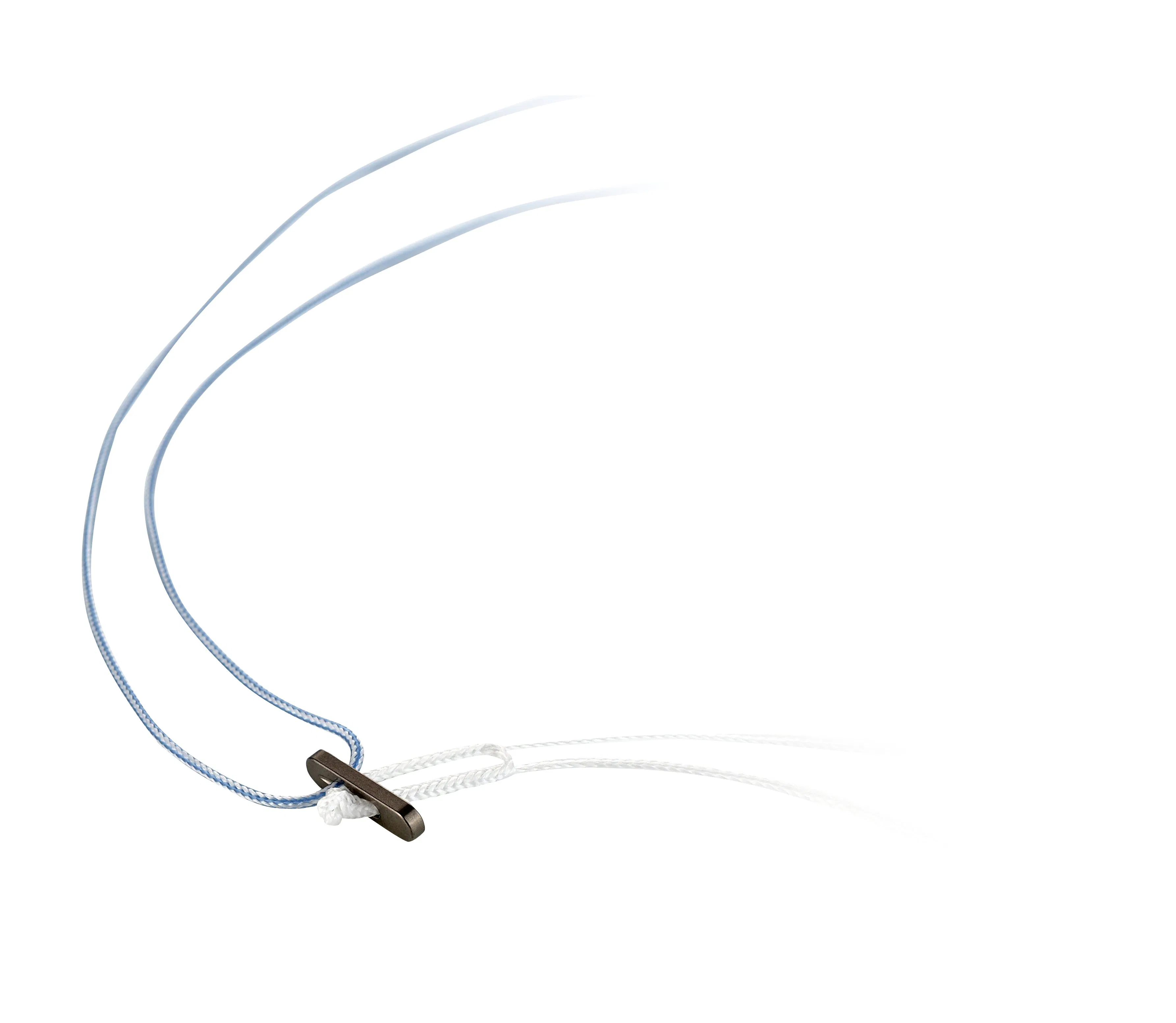
High Quality CE ISO Certified Cruciate Ligament Circle Titanium Button
Designed for cruciate ligament reconstruction, providing high-strength suspension fixation and extensive coverage of cortical bone. Available in various sizes for versatile surgical needs.

| Name | Description | Model | Size |
|---|---|---|---|
| Titanium Button with Loop I | Suitable for cruciate ligament reconstruction; Loop can be adjusted freely. | WZ-TB-101 | D: 3.5mm |
| Titanium Button II | Extensive coverage of cortical bone; High-strength suspension fixation. | WZ-TB-201 | D: 8.0mm |
| Revision Titanium Button I | Better suspension fixation; Use with WZ-TB-101. | WZ-TB-202 | D: 3.5mm |
| Circular Titanium Button I | Various sizes available for specific clinical requirements. | WZ-TB-203 | 11.0mm, 14.0mm, 20.0mm |
| Loop | Standardized loop for fixation. | WZ-TB-301 | f(min): 15.0mm |






