
1 / 4
 Grai Orthopedic Implants
Grai Orthopedic Implants








| Customization: | Available |
|---|---|
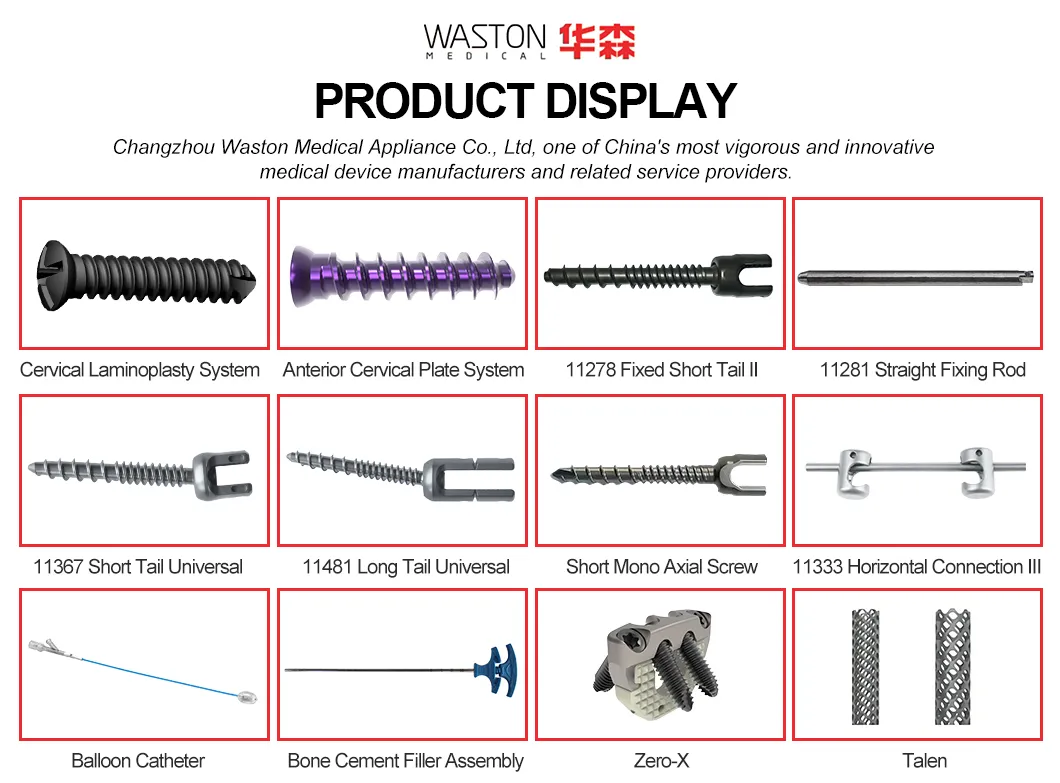
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
| PRODUCT NAME | PRODUCT CODE | DIA | DATA (mm) |
|---|---|---|---|
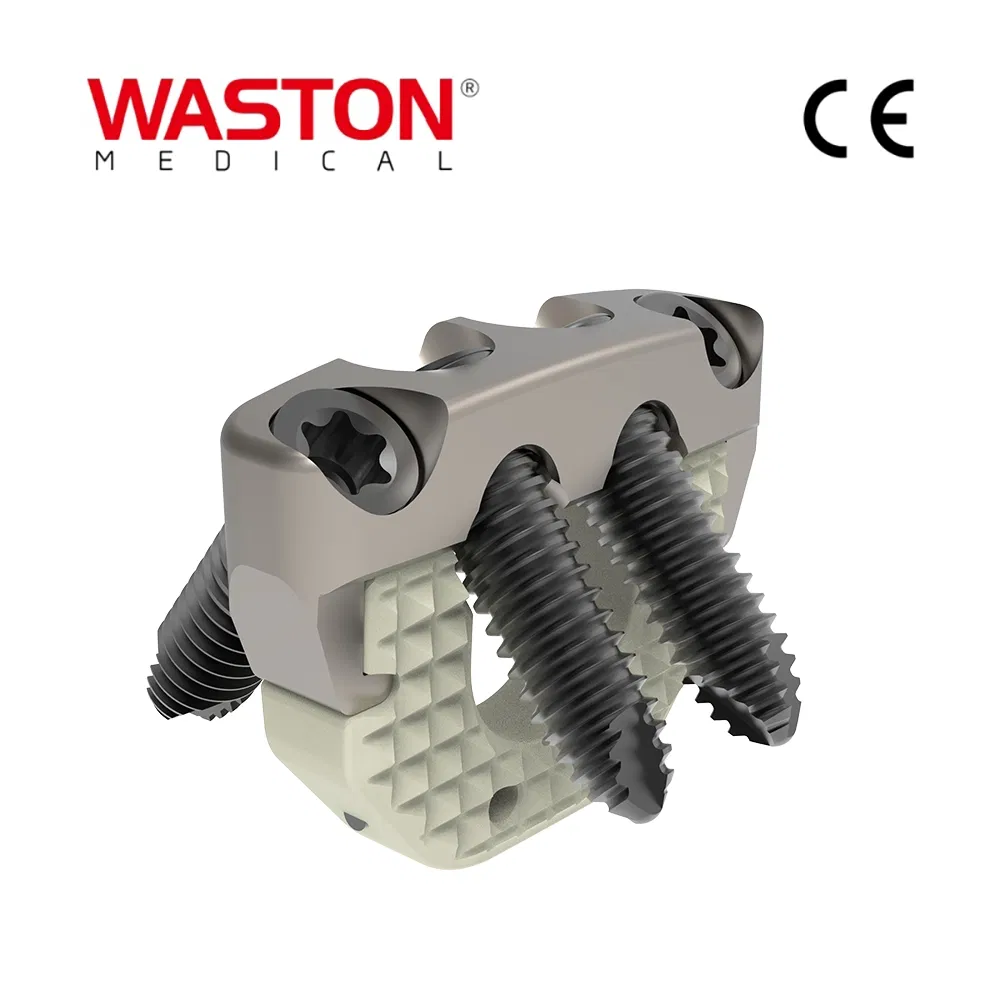
| NEULEN Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| NEULEN Screw (Cross self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | L=8 | ||
| 10511310 | L=10 |


Our facility operates strictly according to the ISO13485 quality system. We implement medical device manufacturing quality management, implants and sterile medical device execution rules, as well as CE MDD 93/42/EEC directive implementation.
Equipped with high-end processing machinery including machining centers from Germany and USA, and automatic lathes from Japan, we ensure first-class quality. The testing center features a biomechanical laboratory and physical-chemical testing center for rigorous raw material and product quality control.

Products are exported to many countries including France, Turkey, Indonesia, Mexico, Russia, and Thailand. Feedback from these regions confirms that our quality meets and exceeds local standards.


