1 / 5
 Grai Orthopedic Implants
Grai Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |

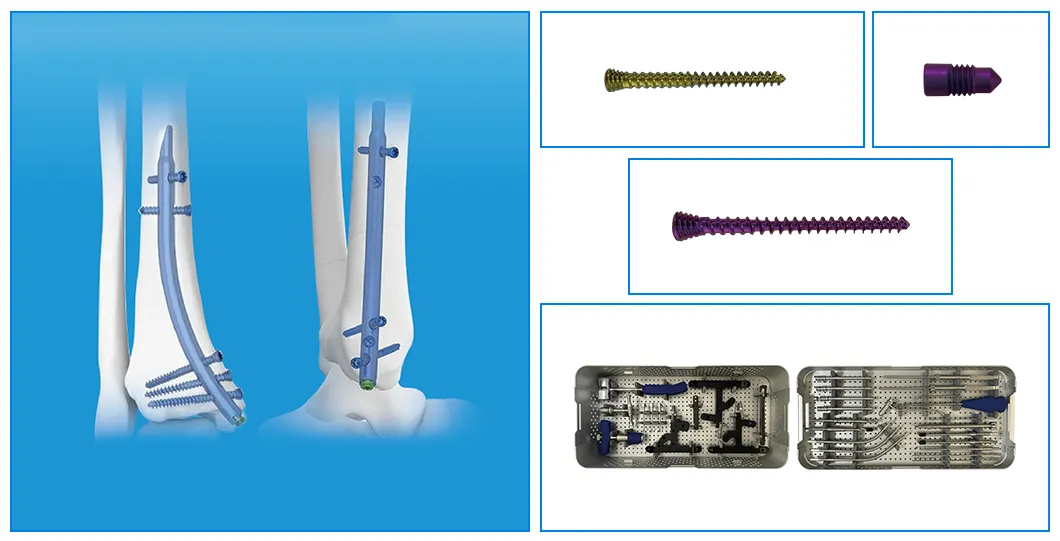
| Product | Size / Length (mm) | Material |
|---|---|---|
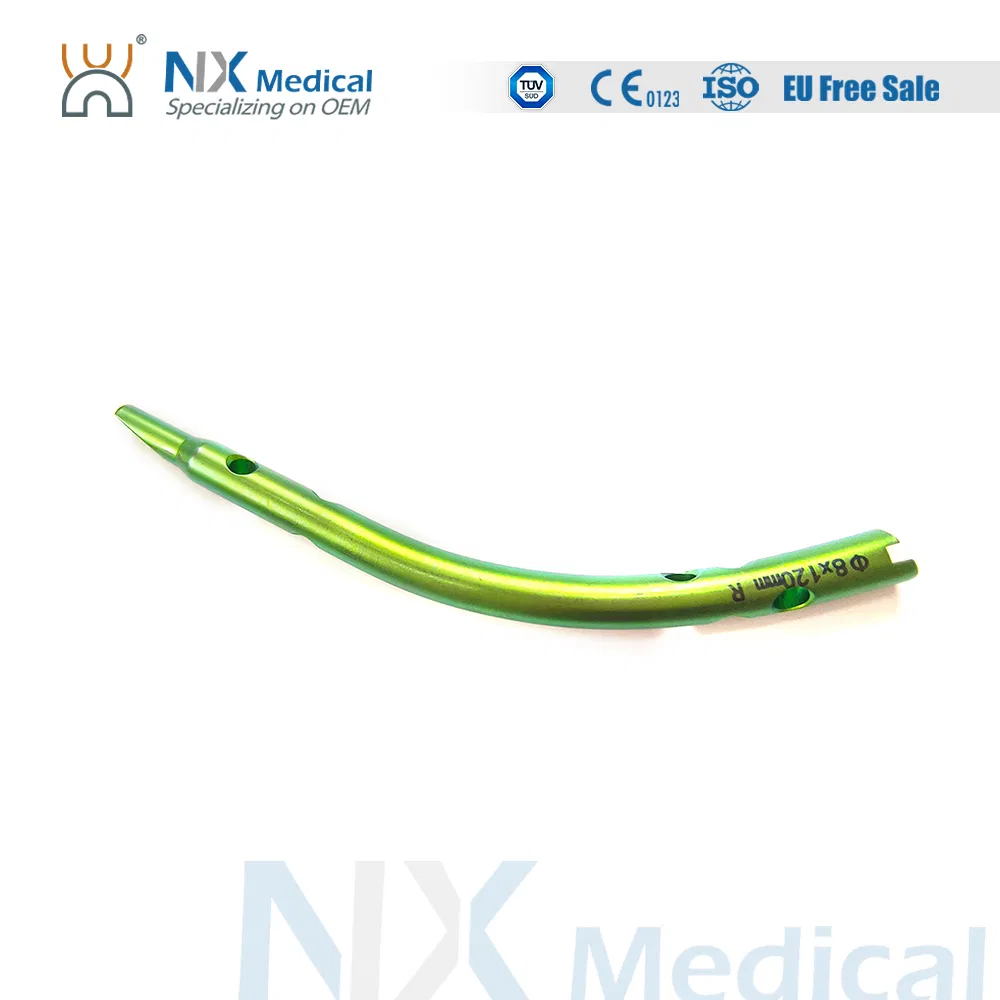
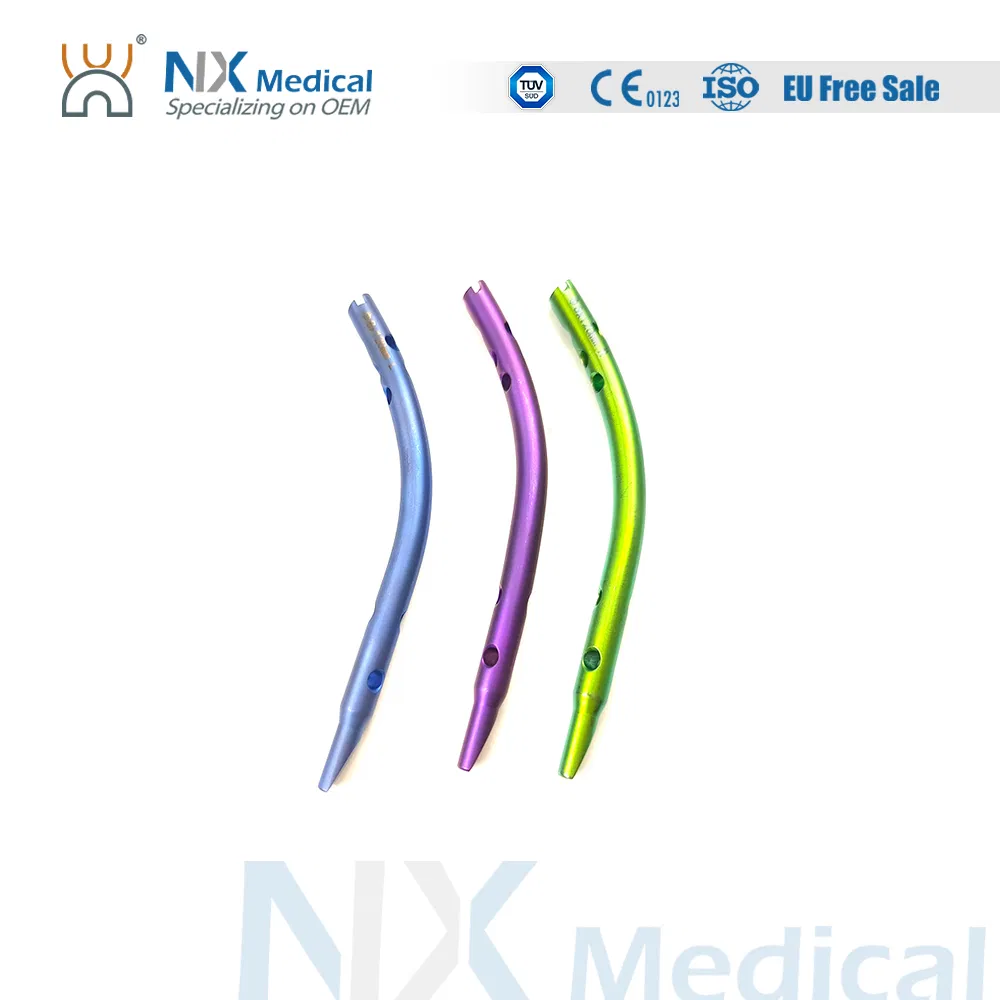
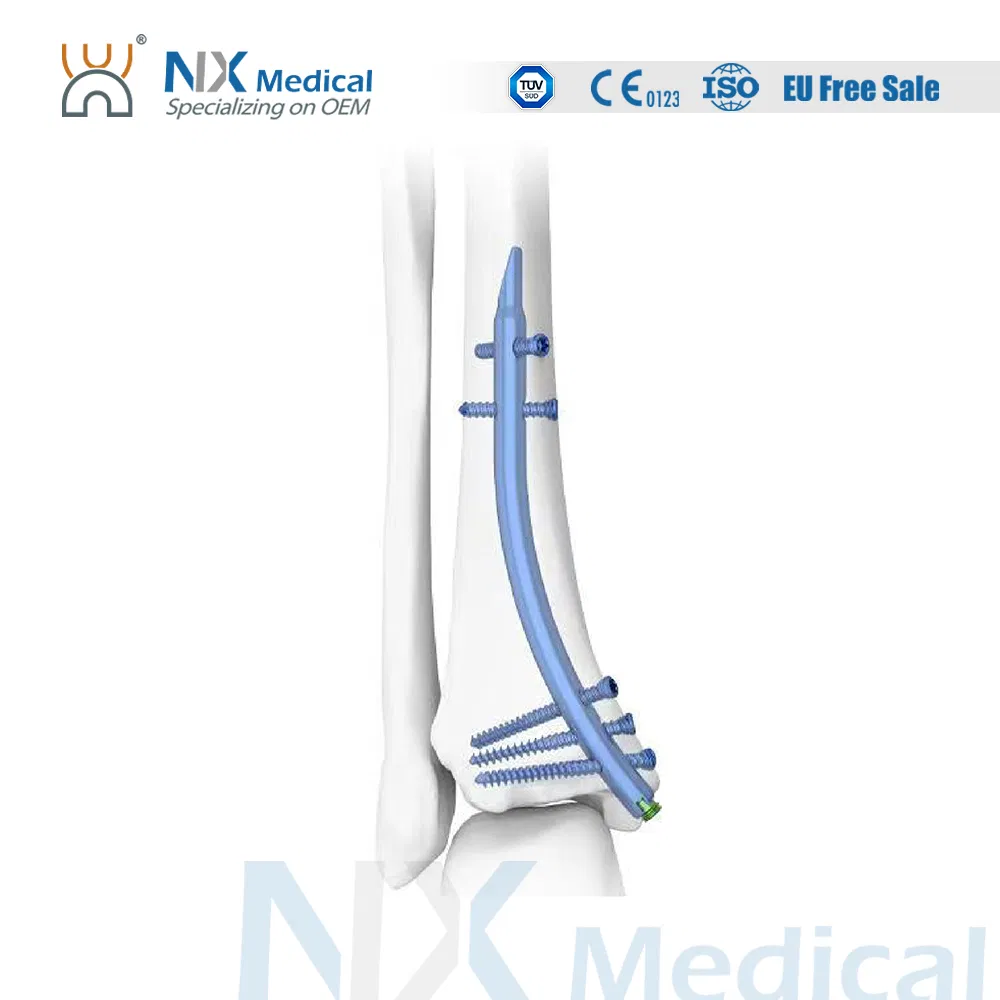
| Retrograde Tibial Nail | 7 x 110 / 8 x 120 / 8 x 140 | Titanium Alloy |
| Interlocking Cancellous Screw (φ3.5mm) | 30 - 52 (2mm increments) | Titanium Alloy |
| Interlocking Cortical Screw (φ3.5mm) | 18 / 20 / 22 / 24 / 26 / 28 | Titanium Alloy |
| Interlocking Cortical Screw (φ4.0mm) | 24 / 26 / 28 / 30 / 32 / 34 | Titanium Alloy |
| Nail End Cap | Standard / 5mm | Titanium Alloy |
Our facility provides a comprehensive range of cost-effective surgical and reliable therapeutic solutions. We offer various kinds of orthopedic trauma implants, spinal implants, and surgical instruments worldwide, committed to the global industry through continuous investment in R&D.



Superior quality is the key for patients using orthopedic products. We have established a strict quality system for frequent self-audit. Externally, we comply with ISO13485, and all products are certified by TUV CE0123 and NMPA. Our goal is to provide the safest and most advanced implants ensuring rapid recovery and a pain-free life.