




1 / 5
 Grai Orthopedic Implants
Grai Orthopedic Implants










| Customization: | Available |
|---|---|
| Customized: | Customized |
| Certification: | ISO, CE |
| Condition | New |
|---|---|
| Usage | Fracture Fixation |
| Using Site | Posterior Spinal Internal Fixation |
| Type | Internal Fixation |
| Model Number | GB1z-III |
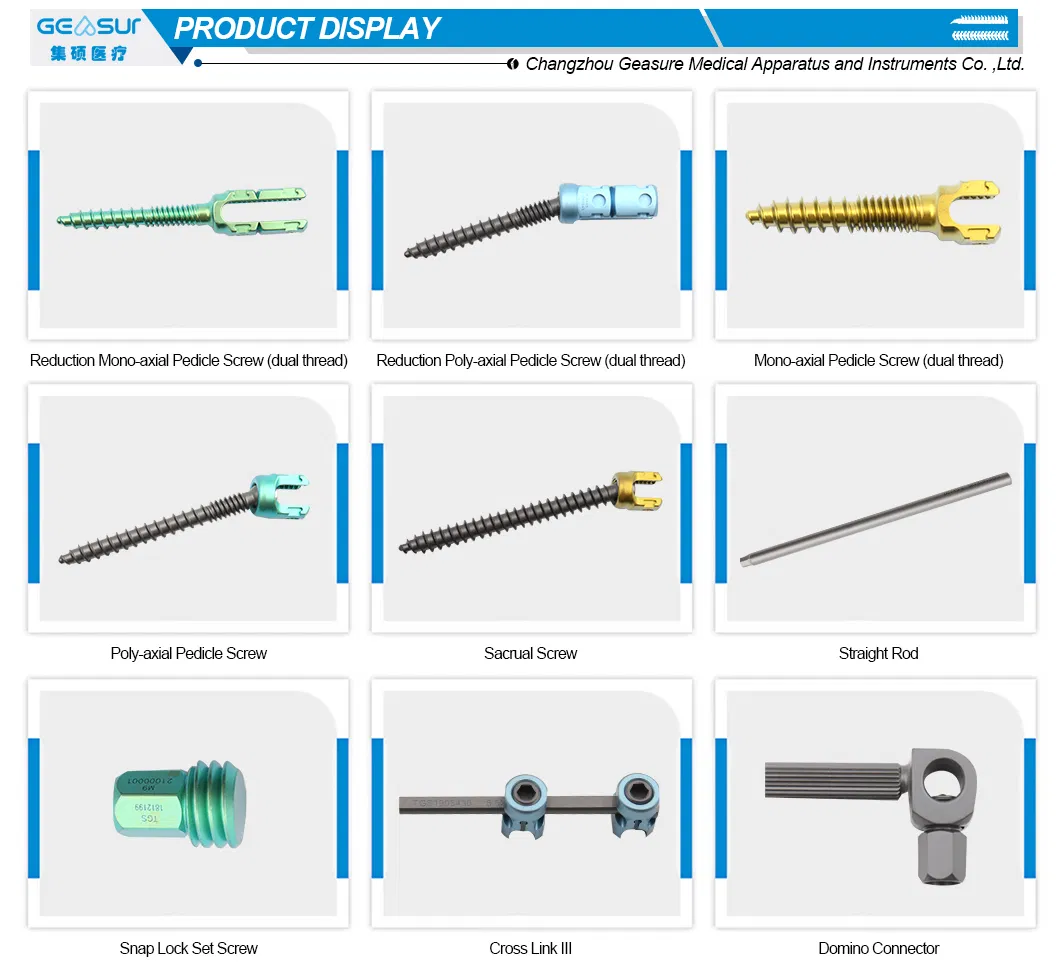
| Product Name | Domino Connector |
| Material | Titanium Alloy |
| Instrument Classification | Class III |
| Warranty | Lifetime |
| Transport Package | Standard Export Packaging |
| Origin | China |




| Posterior Spinal Internal Fixation 5.5 System | |||
|---|---|---|---|
| Domino Connector (Material: Titanium) | |||
| Code (non sterile) | Code (sterile) | Diameter (mm) | Length (mm) |
| 21005510 | 21085610 | 5.5 | 10 |
| 21005520 | 21085620 | 5.5 | 20 |
| 21005530 | 21085630 | 5.5 | 30 |

Established in October 2015, we specialize in the research and production of orthopedic implants and instruments, including trauma, spine, joint, and sports medicine. Our products serve markets globally, supported by first-class production equipment and professional R&D teams.
We operate an advanced orthopedic laboratory in collaboration with university professors and surgeons to ensure clinical excellence. Our quality management systems are ISO13485 and CE certified, with several products registered under FDA 510k.
Core Philosophy: Quality is the foundation of life & Innovation is the road of development.



