



1 / 5
 Grai Orthopedic Implants
Grai Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 66/Piece Request Sample |
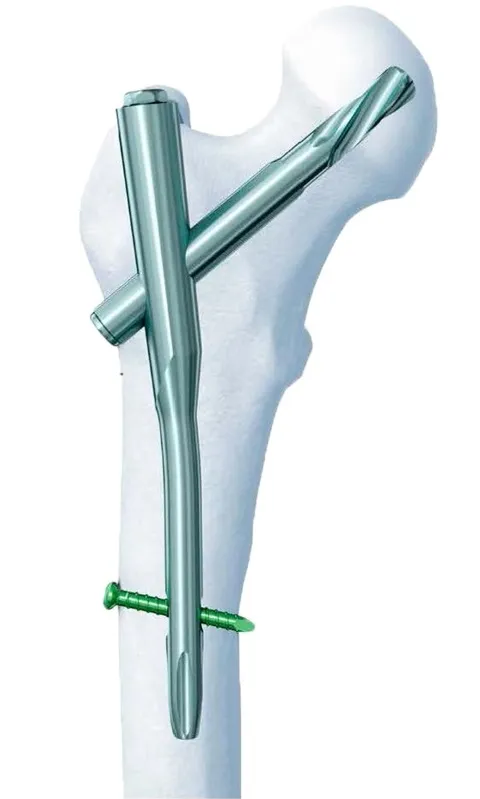
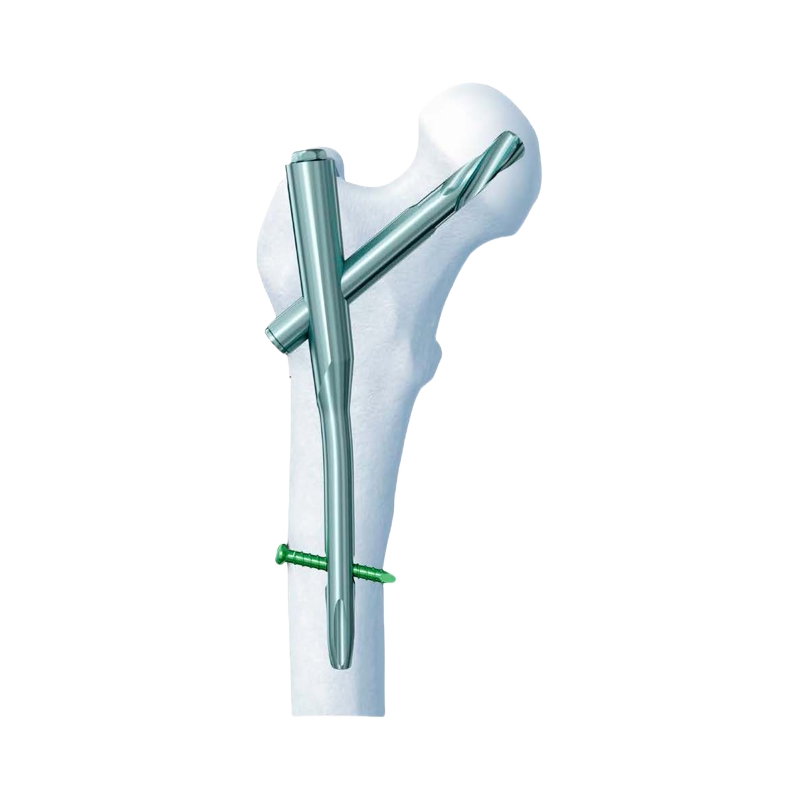
PFNA Standard: Intertrochanteric fracture(31-A1, 31-A2), Reverse intertrochanteric fracture (31-A3), High position Subtrochanteric fracture.
PFNA Long: Low and extended subtrochanteric fractures, Ipsilateral trochanteric fracture, Multiple fractures in the proximal femur, Pathological fracture.
| Product Name | Specification |
|---|---|
| Proximal Femoral Nail | Diameter: 9.0/10.0/11.0/12.0mm, Angle: 130°, Length: 170-240mm |
| Proximal Femoral Nail, Long | Diameter: 9.0/10.0/11.0mm, Angle: 130°, Left/Right, Length: 320-420mm |
| Helical Blade | Diameter: 10.5mm, Length: 70-120mm |
| Locking Bolt | Diameter: 4.9mm, Length: 30-72mm |
| End Cap | Diameter: 10mm, Length: 15-35mm |




Packing Details:
Delivery Way:

Our production base covers an area of 30 thousand square meters, equipped with high-end CNC machining equipment from the United States and advanced quality detection equipment from Japan. We focus on providing one-stop orthopedic product solutions including spinal series, metallic bone plates, and intramedullary nails.

