




1 / 5
 Grai Orthopedic Implants
Grai Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 48/Set Request Sample |

| Name | Number | Size | Material |
|---|---|---|---|
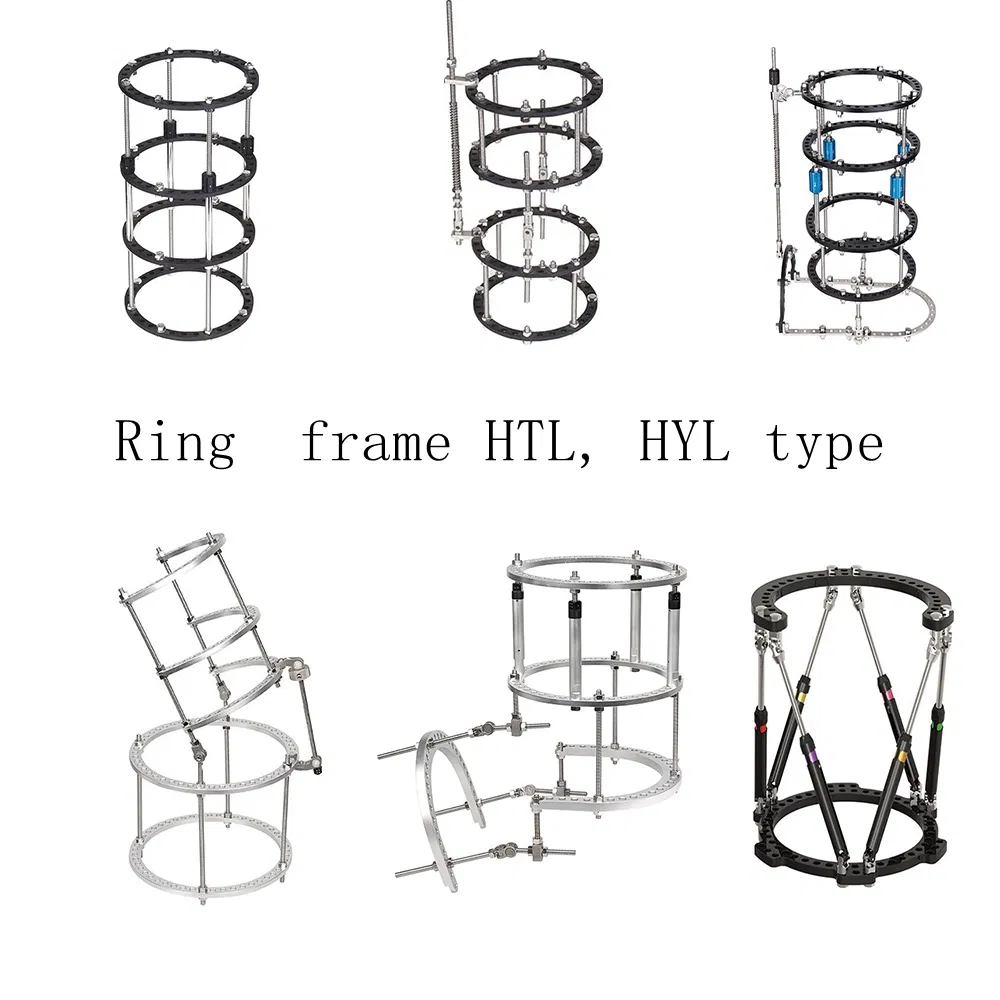
| Half ring (4 ears) | HTLL0301 | L | Aluminum |
| HTLL0302 | M | ||
| HTLL0303 | S |


Established in 1958, we specialize in the development, manufacture, and sales of orthopedic implants and instruments. Our comprehensive product line covers spine pedicle screws, interlocking nails, locking plates/screws, trauma plates, cannulated screws, external fixators, hip prosthesis, and specialized surgical instruments.
Equipped with advanced imported CNC machines and high-precision digital control systems, our production facilities include a 100,000 Grade axenic purification chamber to ensure superior product quality. We are committed to innovative technology and providing complete clinical solutions for bone fracture fixation and deformity correction.


