




 Grai Orthopedic Implants
Grai Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |
Precision Implantation Suture Anchor System for Soft Tissue Surgery



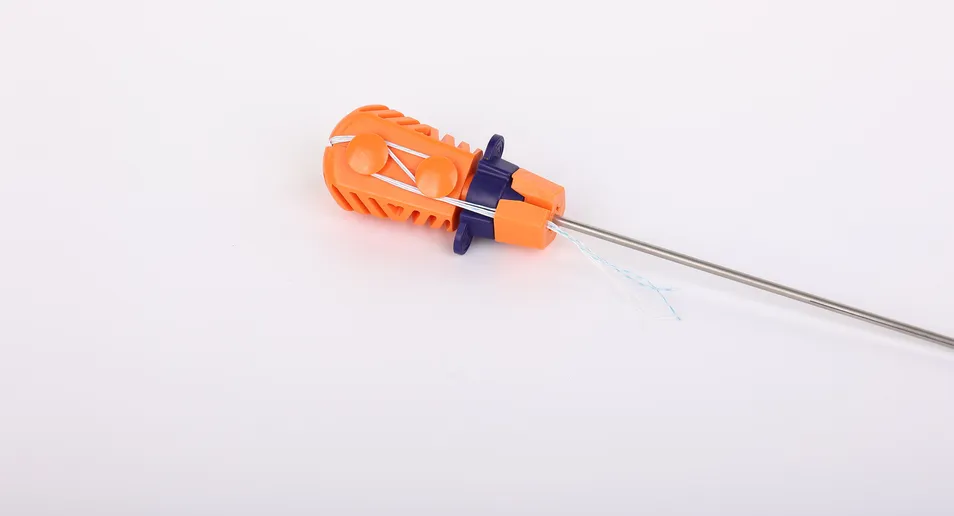
Anchor: Made of high-grade Ti6Al4V alloy, conforming to ISO5832-3:2016 standards for biocompatibility and strength.
Suture: Constructed from ultra-high molecular weight polyethylene (UHMWPE) without coating. Woven from several UHMWPE yarns, it meets ASTM F2848-17 requirements. The blue suture is dyed with FDA-approved materials (Code 73.1015).
Inserter: The contact part is made of medical-grade stainless steel (ASTM F899-12b), while the handle features ergonomic polycarbonate and ABS material.

Our orthopedic surgical consumables are manufactured in Class 100,000 clean workshops supported by Class 10,000 purification laboratories. This ensures the highest safety standards during the R&D and manufacturing cycles.


