




1 / 5
 Grai Orthopedic Implants
Grai Orthopedic Implants










| Customization: | Available |
|---|---|
| Customized: | Customized |
| Certification: | ISO, CE |




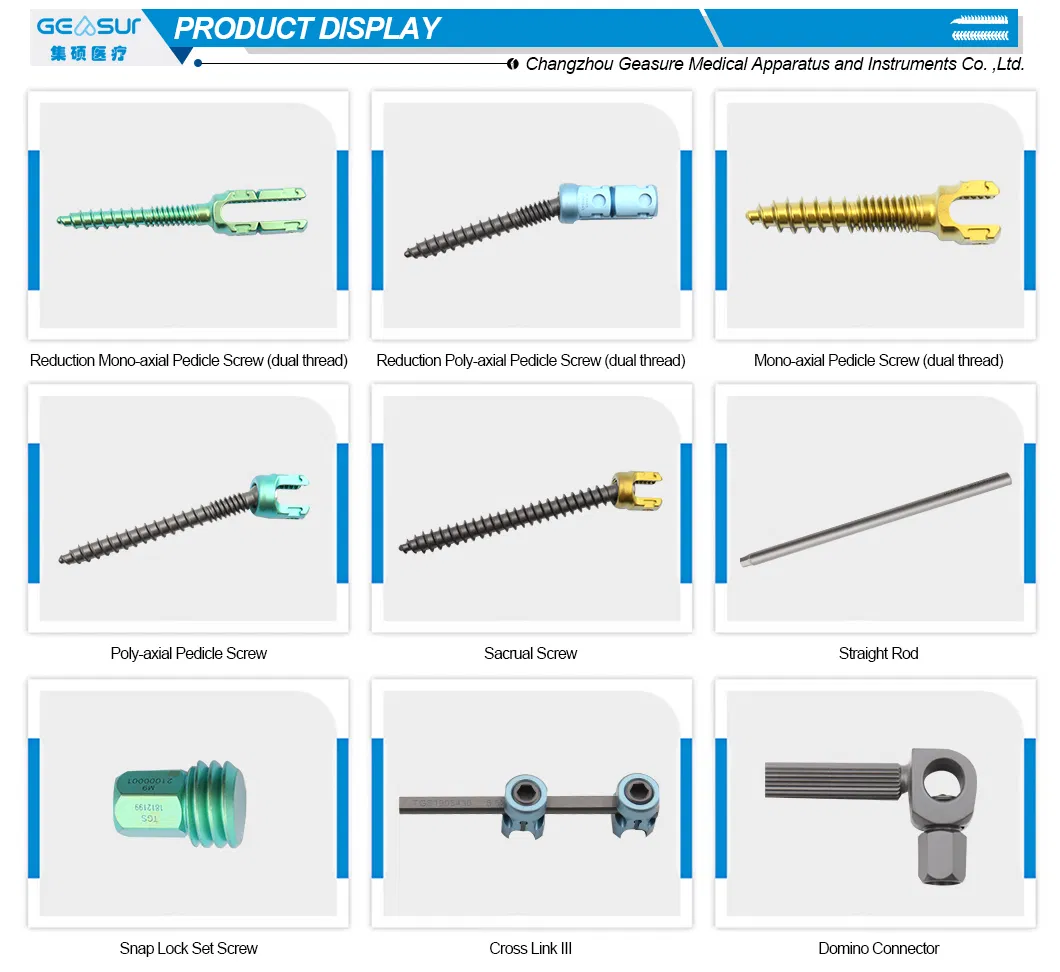
| Posterior Spinal Internal Fixation 5.5 System - Domino Connector | |||
|---|---|---|---|
| Cod(non sterile) | Cod(sterile) | Diameter (mm) | Length (mm) |
| 21005510 | 21085610 | 5.5 | 10 |
| 21005520 | 21085620 | 5.5 | 20 |
| 21005530 | 21085630 | 5.5 | 30 |

The company was established in October 2015, specializing in the research and production of orthopedic implants and instruments, including trauma, spine, joint, and sports medicine. The sales market covers the entire domestic market and many international regions.
We feature first-class production equipment and a professional R&D and manufacturing team. We have established an orthopedic laboratory collaborated with university professors and surgeons to provide operational demonstrations.
We have obtained ISO13485 system certification and CE certification. Some products have been registered with the FDA 510k. Our goal remains: "Quality is the foundation of life & Innovation is the road of development".



