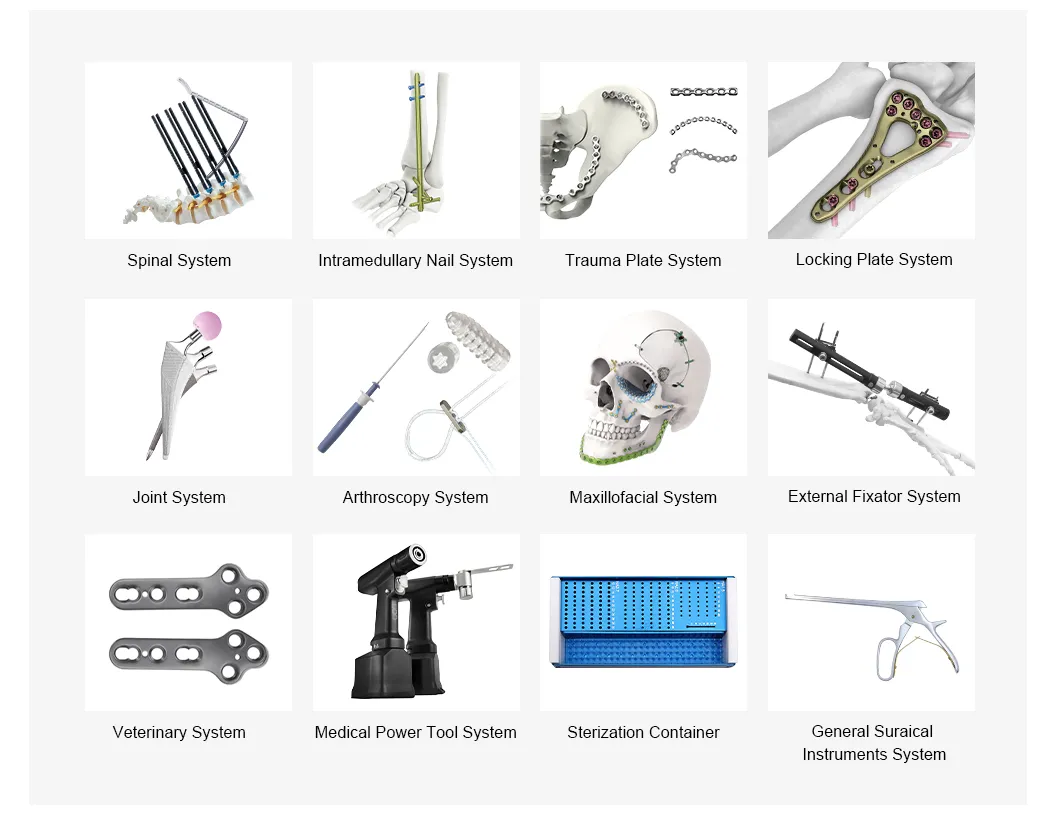


 Grai Orthopedic Implants
Grai Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 413/Piece Request Sample |
| Model NO. | 2303.2410201 |
| Material | Titanium |
| Feature | Disposable |
| Certification | CE, FDA, ISO13485 |
| Group | Adult & Children |
| Product Name | Non-Absorbable Suture Anchor |
| Shipping | DHL/UPS/FedEx/TNT/EMS |
| MOQ | 1 Piece |
| OEM | Available |
| Package | PE Inner Bag + Carton |
| Origin | China |
| HS Code | 9021100090 |


Modern implants utilize advanced bearing surfaces like ceramic-on-ceramic or ceramic-on-polyethylene, which are highly wear-resistant. This greatly extends the implant's lifespan.
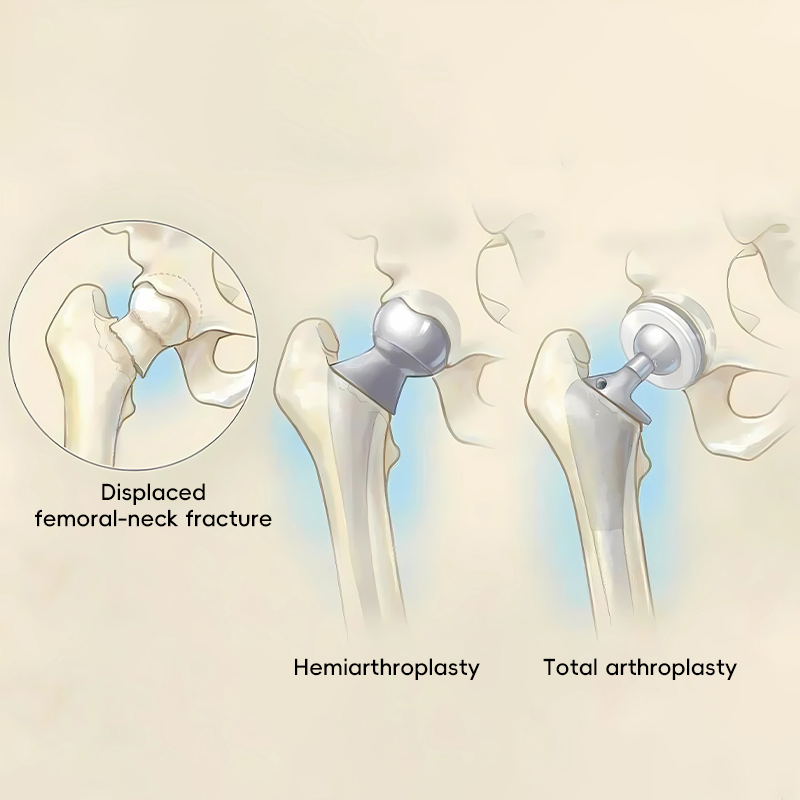
Covers both hemiarthroplasty and total hip replacement, allowing surgeons to choose the best path for patient recovery.

Through a modular model design system, surgeons can tailor the procedure for each patient like assembling parts for a custom fit.
Clear matching lists define which models of femoral heads, stems, and cups can be safely combined for maximum safety.


| Hip Joint System | Introduction |
|---|---|
| Acetabular Cup | Implanted into the pelvis to replace damaged cartilage. Typically porous for osseointegration. |
| Acetabular Liner | Wear-resistant insert (polyethylene/ceramic) that serves as the actual bearing surface. |
| Femoral Head | Spherical component that rotates within the liner, enabling full range of motion. |
| Femoral Stem | Rod inserted into the femur for stability and structural support of the femoral component. |
As a leader in orthopedic implants and instruments manufacturing, we have been successfully supplying clients in 70+ countries for over 15 years.

We provide a 1-year warranty. During the warranty period, we will repair or replace products for free. All inquiries receive responses within 2 days.