 Grai Orthopedic Implants
Grai Orthopedic Implants
 Grai Orthopedic Implants
Grai Orthopedic Implants
 Orthopedic Intertan Femoral Nail Instrument Set (Type 1) Surgical Instrument Medical Equipment
Orthopedic Intertan Femoral Nail Instrument Set (Type 1) Surgical Instrument Medical Equipment
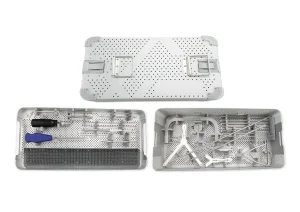 Orthopedic Surgery Osteotomy Instrument Set Trauma Instrument
Orthopedic Surgery Osteotomy Instrument Set Trauma Instrument
 CE Certified Orthopedic Expert Femoral Instrument Set for Shoulder Surgery Essential Orthopedic Surgical Instruments
CE Certified Orthopedic Expert Femoral Instrument Set for Shoulder Surgery Essential Orthopedic Surgical Instruments
 Replacement CE Sterilized, Carton Package Prosthesis Knee Joint
Replacement CE Sterilized, Carton Package Prosthesis Knee Joint
 Prosthesis Replacement Sterilized Package Class III Knee Joint
Prosthesis Replacement Sterilized Package Class III Knee Joint
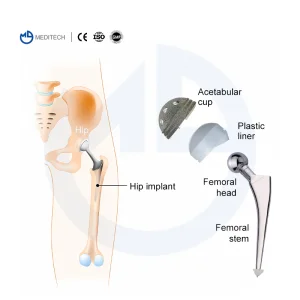 Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
 Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China
Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China
 Total Hip Replacement Prosthesis Corail Femoral Stem, Artificial Hip Joint System Titanium Hip Joint Prosthesis Implant
Total Hip Replacement Prosthesis Corail Femoral Stem, Artificial Hip Joint System Titanium Hip Joint Prosthesis Implant
The global orthopedic market is witnessing a significant shift towards Revision Arthroplasty. As primary joint replacements performed in previous decades reach the end of their lifespan, the demand for sophisticated Revision Hip/Knee Systems has skyrocketed. Revision surgery is inherently more complex than primary replacement, requiring specialized implants that can address bone loss, instability, and fractured previous components. Leading manufacturers and factories worldwide are now focusing on modularity and biological fixation to ensure better clinical outcomes for an aging global population.
With an increasing life expectancy and more active elderly populations, the frequency of secondary surgeries is rising. Key drivers include implant wear, aseptic loosening, and periprosthetic infections, necessitating high-performance revision systems.
Factories are integrating 3D printing (additive manufacturing) to create porous titanium structures that mimic human bone. This enhances osseointegration and provides better long-term stability for revision implants.
The industrial landscape is moving towards a "Quality-First" approach. Modern factories in China are now competing globally by adhering to MDR (Medical Device Regulation) and FDA standards while maintaining competitive production scales.
As a leading Chinese orthopedic factory, we combine high-volume production with surgical-grade precision. Our facility is designed to meet the rigorous demands of global healthcare providers, offering a comprehensive suite of revision and trauma solutions.
Our factory is certified with ISO13485 and BSCI. Every product, from the S-ROM Titanium Hip Joint to the Total Knee Prosthesis, undergoes strict raw material traceability and comprehensive laboratory testing. We are proud to hold CE, MDR, and FDA certifications, ensuring our products are ready for use in any medical environment worldwide.

Modern procurement departments in hospitals and medical distributors are no longer just looking for the lowest price; they are seeking long-term reliability and innovative surgical compatibility. The global trend is moving towards integrated systems—where the implant and the instrument set are perfectly synchronized to reduce surgical time and improve accuracy.
Revision surgeries often present unpredictable bone defects. Procurement teams prioritize systems like the S-ROM titanium alloy cementless hip joints, which allow for intraoperative flexibility. Modular stems and acetabular cups enable surgeons to customize the fit to the patient’s unique anatomy on the fly.
The shift from traditional stainless steel to high-strength titanium alloys and UHMWPE (Ultra-High-Molecular-Weight Polyethylene) inserts has defined the new era of longevity. These materials reduce wear debris and minimize the risk of osteolysis, which is the leading cause of revision failure.
Factories are now aligning their production with robotic-assisted surgery platforms. This requires high-precision manufacturing where tolerances are measured in microns, ensuring that every femoral stem and knee component fits perfectly within the digitally planned surgical space.

China has emerged as a global hub for medical device manufacturing, combining advanced technology with unparalleled supply chain efficiency. Our factory exemplifies these advantages:
Our experienced R&D team, including 8 senior engineers, introduces 10 new products every month. This allows our partners to stay ahead of market trends and offer the latest surgical solutions to their clients.
By optimizing manufacturing processes and leveraging local material supplies, we provide premium Revision Hip/Knee Systems at a fraction of the cost of Western competitors, without compromising on CE/FDA standards.
We offer free replacements within one year and dedicated after-sales support. Our agency policy assistance helps distributors establish a strong local presence quickly.
Revision Hip and Knee systems are used across diverse clinical settings globally:
 S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
 Disposable Pulsed Lavage System
Disposable Pulsed Lavage System
 Surgical Dtn Retrograde Tibia Nail Instrument
Surgical Dtn Retrograde Tibia Nail Instrument
 Fns Femoral Neck System Instrument Set
Fns Femoral Neck System Instrument Set
 CE Approved Replacement Sterilized Package Prosthesis Knee Joint
CE Approved Replacement Sterilized Package Prosthesis Knee Joint
 Advanced Hip & Knee Prosthesis with Durable Materials
Advanced Hip & Knee Prosthesis with Durable Materials
 Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Ortho Surgery Arthroplasty Tka
Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Ortho Surgery Arthroplasty Tka
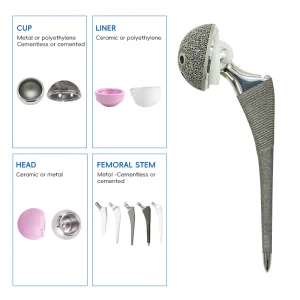 UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System Total Knee Replacement Prosthesis Implants Instruments Set
UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System Total Knee Replacement Prosthesis Implants Instruments Set
Revision Hip/Knee systems are not just "larger versions" of primary implants. They are engineered to solve specific biological challenges. When a primary implant fails due to osteolysis, the surrounding bone is often compromised. Our factory specializes in producing Augmentation Blocks and Metaphyseal Sleeves that provide additional support where bone stock is lacking.
We utilize Ti-6Al-4V ELI titanium alloy for its superior fatigue strength and biocompatibility. Our Cobalt-Chromium-Molybdenum (CoCrMo) femoral components are polished to a mirror finish to ensure the lowest possible coefficient of friction against UHMWPE liners. This meticulous attention to material science is what differentiates our factory-produced components from standard offerings.
With a 15,000 m² production base and a team of 500 professionals, we are equipped to handle large-scale international tenders and specialized hospital orders. Whether you are looking for a reliable OEM manufacturer for Total Hip Replacement systems or a supply partner for advanced Trauma Instrument sets, our facility offers the scale, certification, and technical expertise required in the modern medical market.
In conclusion, the future of Revision Hip/Knee systems lies in the intersection of AI-driven design, robotic surgical compatibility, and biological material innovation. As a premier manufacturer, we are committed to leading this charge, providing surgeons and patients with the tools they need for a more mobile, pain-free life.