 Grai Orthopedic Implants
Grai Orthopedic Implants
 Grai Orthopedic Implants
Grai Orthopedic Implants
 Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants Bone Implant
Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants Bone Implant
 Medical OEM&ODM Titanium Cannulated Headless Compression Screw Hollow Orthopedic Implant Cannulated Screw Bone Screws
Medical OEM&ODM Titanium Cannulated Headless Compression Screw Hollow Orthopedic Implant Cannulated Screw Bone Screws
 Cannulated Headless Compression Herbert Bone Screw Set Titanium Locking Orthopedic Trauma Surgical Implants Screws Price
Cannulated Headless Compression Herbert Bone Screw Set Titanium Locking Orthopedic Trauma Surgical Implants Screws Price
 Carbon Steel4.8/8.8/10.9 Fastener Flower Countersunk Head Self-Tapping Screws for Kitchen/Cabinet/Furniture
Carbon Steel4.8/8.8/10.9 Fastener Flower Countersunk Head Self-Tapping Screws for Kitchen/Cabinet/Furniture
 SS304 Carbon Steel Knurled Head Thumb Screw
SS304 Carbon Steel Knurled Head Thumb Screw
 Customizable Steel Steel Black Finish Knurled Thumb Screw with Washer
Customizable Steel Steel Black Finish Knurled Thumb Screw with Washer
 Carbon Steel/4.8/8.8/10.9 M2.5/Galvanized/Customized Double Rod Fastener Knurled Bamboo Screw
Carbon Steel/4.8/8.8/10.9 M2.5/Galvanized/Customized Double Rod Fastener Knurled Bamboo Screw
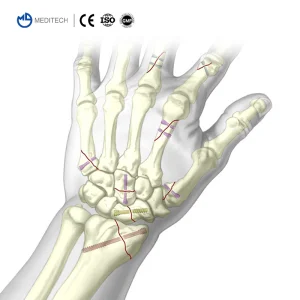 Titanium Medical Bone Screws Herbert Cannulated Compression Screw Price
Titanium Medical Bone Screws Herbert Cannulated Compression Screw Price
The global orthopedic market has witnessed a paradigm shift toward minimally invasive surgeries (MIS), where Hollow Bone Screws, also known as cannulated screws, play a pivotal role. These specialized fasteners are designed with a central hollow core, allowing them to be guided over a K-wire for extreme precision in fracture fixation. As a leading China Hollow Bone Screws manufacturer, we recognize that the demand is no longer just for basic stabilization but for biomechanical excellence that accelerates patient recovery.
In today's commercial landscape, the industry is valued at billions of dollars, with a compound annual growth rate (CAGR) driven by an aging global population and an increase in sports-related injuries. North America and Europe remain the largest consumers, but the Asia-Pacific region is the fastest-growing market due to expanding healthcare infrastructure and the presence of high-tech manufacturing hubs in China. Modern hospitals and surgical centers now prioritize implants that offer both titanium biocompatibility and mechanical strength to minimize secondary surgeries.
Utilization of Medical Grade Titanium (Ti-6Al-4V ELI) and Stainless Steel for optimal bone integration.
Adherence to global standards including FDA 510(k) clearances and CE MDR certifications for safe clinical use.
Advanced CNC machining processes ensuring thread accuracy within microns for superior pull-out resistance.

Our flagship Headless Compression Hollow Screws represent the pinnacle of orthopedic fixation technology. Designed specifically for small bone fractures and arthrodesis, the headless design ensures zero prominence above the bone surface, significantly reducing soft tissue irritation.
Key Benefits Include:
The industry is moving toward "Smart Implants" and bio-resorbable materials. However, the current gold standard remains Titanium Cannulated Screws due to their proven track record in osseointegration. A significant trend is the customization of screw geometries for specific anatomical sites—such as the scaphoid, femoral neck, or tibial plateau. Another trend is the integration of anti-microbial coatings to prevent surgical site infections (SSI), a top priority for global healthcare providers.
Digitalization in manufacturing, specifically the use of AI-driven quality control and 3D simulation of screw-to-bone interaction, is setting a new benchmark for bone screw suppliers. As a technology-forward factory, we utilize these advancements to ensure our products meet the "zero-failure" requirement of modern trauma centers.
Procurement departments from multinational medical device brands (OEM/ODM) look for three critical factors: Quality Compliance, Supply Chain Stability, and Cost-Efficiency. China has evolved from a low-cost manufacturing hub into a high-tech center for medical devices. Our factory exemplifies this transition by offering:
Factory covers an area of 15,000 m², Factory certified with ISO13485, and BSCI. Products certified with CE, MDR, and FDA. Our team consists of 500 employees, including 8 senior engineers and 10 QC specialists. We offer Minor Customization, Raw material traceability identification, and rigorous semi-product and finished product inspections.
We implement strict quality control processes and advanced laboratory facilities for comprehensive testing, ensuring every hollow bone screw leaving our facility is surgical-grade.
Furthermore, our ability to introduce 10 new products every month through our experienced R&D team allows our partners to stay ahead of market trends. We offer a Free replacement within one year and robust after-sales support, bridging the gap between offshore manufacturing and local reliability.
Our Titanium Cannulated Headless Compression Screws are the preferred choice for orthopedic surgeons worldwide. By utilizing high-grade Ti-6Al-4V, we ensure maximum strength-to-weight ratio and excellent biocompatibility, reducing the risk of implant rejection.
Applications Include:

The versatility of cannulated hollow screws allows them to be applied in various clinical scenarios across the globe:
Used for Lisfranc injuries, calcaneal fractures, and hallux valgus corrections where precision in small bones is vital.
Miniature hollow screws provide the stability needed for growth-plate-conscious fixation in young patients.
Essential for hip fracture fixations (femoral neck) where minimally invasive entry reduces post-operative complications for elderly patients.
We believe that "Quality is the lifeblood of medical devices." Our facility integrates advanced robotics with human expertise to deliver 100% compliant medical implants.


With over 300 different models available, we provide a comprehensive portfolio for any trauma or orthopedic need. Our R&D team works closely with clinical advisors to iterate designs based on real-world surgical feedback. Whether it's a specific thread pitch or a unique head geometry, we provide customizable designs and packaging to meet your specific regional regulatory needs.
 Stainless Steel Bone Fixator for Orthopedic Surgery
Stainless Steel Bone Fixator for Orthopedic Surgery
 Low Price Thread Hollow Screw, Titanium Orthopedic Implants, Trauma, Cannulated Screws for Fixation
Low Price Thread Hollow Screw, Titanium Orthopedic Implants, Trauma, Cannulated Screws for Fixation
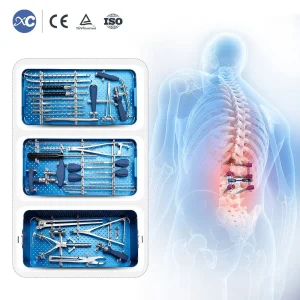 Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle Screw System Instrument Set
Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle Screw System Instrument Set
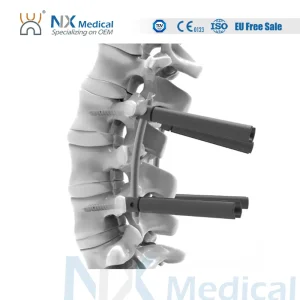 Nx Medical Titanium Cannulated Bone Cement 5.5mm System Pedicle Screw Bone Fixation
Nx Medical Titanium Cannulated Bone Cement 5.5mm System Pedicle Screw Bone Fixation
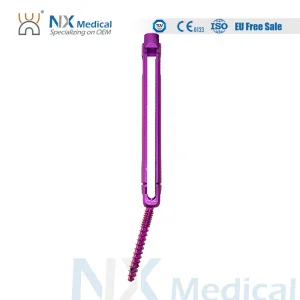 Nx Medical Titanium Cannulated Bone Cement Pedicle Screw Bone Internal OEM ODM
Nx Medical Titanium Cannulated Bone Cement Pedicle Screw Bone Internal OEM ODM
 Orthopedic Implant Cannulated Headless Compression Screw - II Metallic Bone Screws
Orthopedic Implant Cannulated Headless Compression Screw - II Metallic Bone Screws
 5.5/6.5 Orthopedic Series Spinal System Pedicle Spinal Screws
5.5/6.5 Orthopedic Series Spinal System Pedicle Spinal Screws
 High Quality Orthopedic Titanium Metal Cancellous Cannulated Screws Stainless Steel Full-Thread Self-Tapping Bone Screw
High Quality Orthopedic Titanium Metal Cancellous Cannulated Screws Stainless Steel Full-Thread Self-Tapping Bone Screw