



 Grai Orthopedic Implants
Grai Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 60/Piece Request Sample |
| Condition | New |
| Usage | Fracture Fixation |
| Type | Internal Fixation |
| Shape | Ball-Headed |
| Specification | Titanium Alloy TC4 |
| Origin | China |
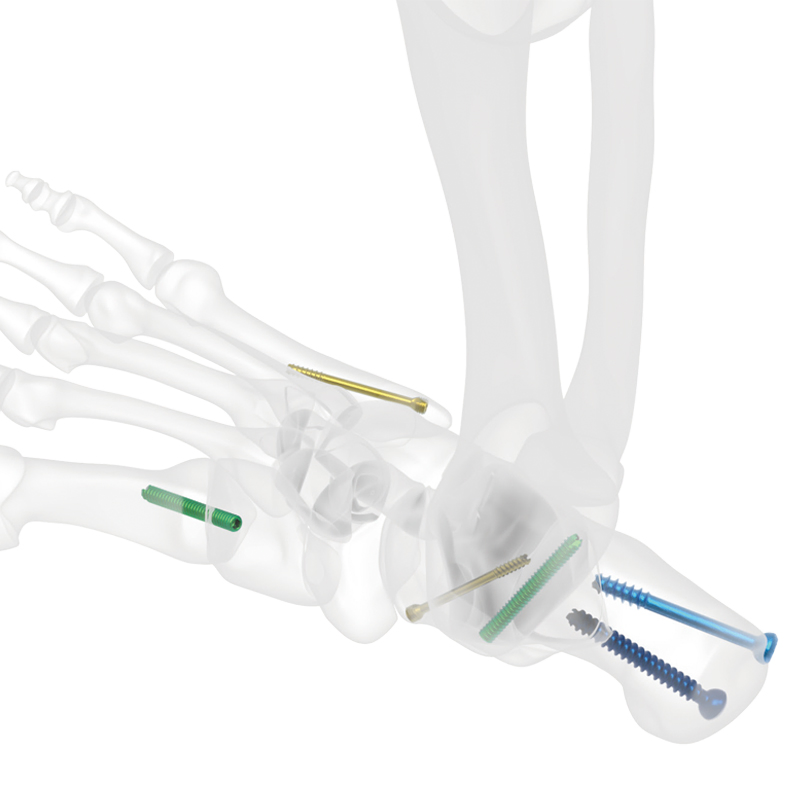
The Ball-Head Cannulated Screw is designed to provide dynamic compression across fracture sites, enhancing friction between bone fragments and between the screw and bone, thereby resisting shear forces and minimizing fracture displacement. The screw features a low-profile ball-head design that minimizes soft tissue irritation while maintaining compatibility with various fracture types.
Its trapezoidal thread structure enhances pressure across the fracture line and significantly increases pullout resistance. Manufactured from high-quality TC4 titanium alloy, the screw also undergoes anodic oxidation to improve biocompatibility, surface hardness, wear resistance, and corrosion resistance. Compatible with two types of washers (Type I and Type II), the system offers secure fixation and reliable load distribution across bone surfaces.
Indicated for internal fixation of fractures in the upper and lower extremities.





